Copy link
Retinoblastoma: Ophthalmic Artery Chemotherapy
Last updated: 04/26/2023
Key Points
- Retinoblastoma (RB) is the most common pediatric intraocular malignancy. The pathognomonic genetic mutation maps to chromosome 13q14, which encodes a tumor suppressor gene (RB1).
- For isolated intraocular RB, selective chemotherapy infusion into the ophthalmic artery (OA) is increasingly used to spare children the morbidity of enucleation and external beam radiation.
- Intra-arterial chemotherapy into the OA can be associated with serious cardiorespiratory complications such as decreased lung compliance, hypotension, and bradycardia.
Epidemiology
- RB is a neuroectodermal tumor and the most common primary pediatric intraocular malignancy (1:20,000 live births) and represents 10-15% of infant malignancies.1 The incidence in males and females is equivocal, as is the incidence across races.2
- RB is classified as heritable (germline mutation) and nonheritable (somatic mutation), with nonheritable types being the most common.2
- Most nonheritable RB cases (60-70%) are unilateral and unifocal and present at a later age than the heritable types (mean age at diagnosis is two years).3
- Heritable RB cases (40%) encompass patients with germline mutations, positive family history, and bilateral or multifocal disease (mean age at diagnosis is one year).3 Children with heritable RB are at increased risk of developing second malignancies like sarcoma.4
Clinical Presentation
- Most RB cases present by five years of age.3,4
- The most common presentation of RB is leukocoria or a white pupillary reflex (Figure 1), which warrants urgent ophthalmologist referral in a child. It can also present as strabismus, nystagmus, and redness.
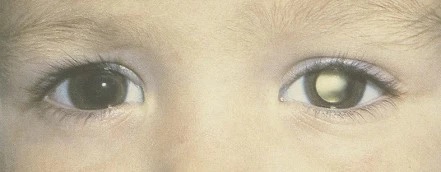
Figure 1. Leukocoria. Source: Aerts I, et al. Retinoblastoma. Orphanet J Rare Dis. 2006.3 CC BY 2.0
- Patients with metastatic disease may present with systemic findings such as weight loss, fatigue, gastrointestinal symptoms, or neurological deficits.
- Ocular fundus exam reveals either single or multiple, whitish-colored masses with associated vessel dilation (Figure 2). There are three main RB growth patterns: exophytic, endophytic, and diffuse infiltrating.5
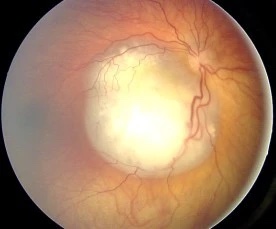
Figure 2. Ocular fundus exam showing whitish mass with angiomatous dilation of vessels. Source: Source: Aerts I, et al. Retinoblastoma. Orphanet J Rare Dis. 2006.3 CC BY 2.0
- Differential diagnosis includes cataracts, Coat’s disease (exudative retinal vascular disorder), persistent fetal vasculature, vitreous hemorrhage, ocular toxocariasis (roundworm infection).3
Screening
- For the general population, screening for RB occurs at pediatrician visits.
- For children with a family history of RB or who have the RB1 mutation, regular screening begins at birth and then continues weekly to monthly, depending on the risk. These exams are typically done by a pediatric ophthalmologist and often require general anesthesia for a comprehensive examination.
- In addition to regular eye examinations, genetic testing, and counseling for RB1 mutation is recommended for patients with a family history of RB.6
Staging
- Disease extent typically involves the following: physical examination, ophthalmologic exam under anesthesia, ocular ultrasonography, optical coherence tomography, and magnetic resonance imaging.
- The International Classification of Retinoblastoma (ICRB) grouping system (Table 1) allows the classification of patients to predict the likelihood of success with chemotherapy and identify patients that can avoid the higher morbidities associated with enucleation and external beam radiation.
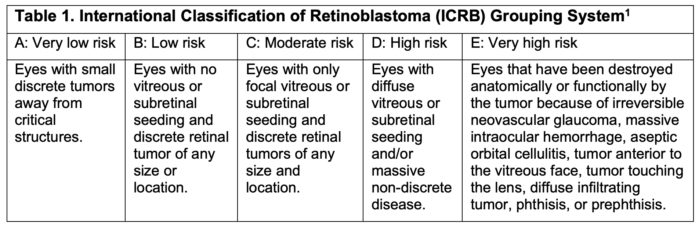
Table 1.
- Patients with enucleation or metastatic spread are staged by the International Retinoblastoma Staging System (IRSS) (Table 2).
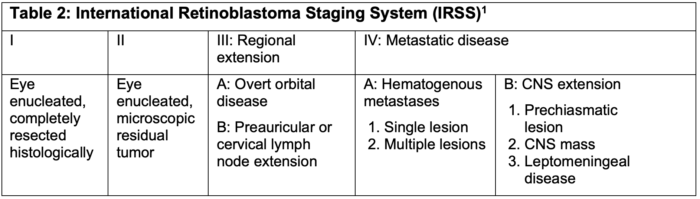
Table 2.
Prognosis
- If untreated, RB is fatal and can invade local structures and metastasize to extraorbital sites. Survival rates with central nervous system disease are low.5
- The US survival rate is greater than 95% due to advances in treatments; eye salvage depends on the stage of disease at diagnosis.3-4
- The most common cause of death in patients with heritable RB is secondary malignancies.4
Treatment
- Treatment options for RB depend on disease severity: chemotherapy (i.e., carboplatin, vincristine, and etoposide), cryotherapy, photocoagulation, thermotherapy, laser ablation, localized radiotherapy, external beam radiation and enucleation.3
- Radiation therapy has been traditionally used to treat retinoblastoma but can cause secondary neoplasms, facial bony deformities, cataracts, and glaucoma; this therapy is often provided during multiple short anesthetics. Cryotherapy and laser therapy can be curative for smaller tumors, but 80% of patients have too large of a tumor burden at presentation.7
- Newer approaches involve localized delivery of chemotherapy via the OA, which can treat the tumor with fewer systemic complications.
Anesthetic Considerations for Selective Chemotherapy to the Ophthalmic Artery
- For this procedure, the femoral artery is cannulated, intravenous heparin is administered, and a microcatheter is advanced into the ophthalmic artery. The chemotherapeutic agents are then administered, sometimes with verapamil to treat/prevent ophthalmic artery spasm. The femoral catheter is then removed, and groin pressure is held for hemostasis. A smooth emergence is desirable to prevent rebleeding from the femoral puncture site.
- Serious adverse cardiorespiratory complications include hypotension, bradycardia, and/or decreased respiratory compliance that mimics acute bronchospasm. They usually occur within 2 minutes after ophthalmic artery catheterization, and deep anesthesia does not reduce the incidence of these complications.8
- This sudden and precipitous decrease in lung compliance occurs in almost one-third of cases and occurs more frequently with subsequent treatments (18% for the first treatment vs. 32% for subsequent treatments).8 Interestingly, there is usually no wheezing or obstructive capnograph and there are generally no respiratory issues in the recovery area.
- If a balloon catheter is used, there can be significant bradycardia (oculocardiac reflex), which can occur despite pretreatment with atropine.
- A topical vasoconstrictor, such as oxymetazoline, is often administered in the ipsilateral nare to reduce loss of chemotherapy through drainage into the external carotid artery.
- A smooth emergence is desirable to prevent rebleeding from the femoral puncture site.
- Postoperative nausea and vomiting prophylaxis is strongly recommended.
- Another postoperative consideration is periorbital edema, especially if more than two chemotherapy agents are given.
References
- Ortiz MV, Dunkel IJ. Retinoblastoma. J Child Neurol. 2016;31(2):227-36. PubMed
- Broaddus E, Topham A, Singh AD. Incidence of retinoblastoma in the USA: 1975-2004. Br J Ophthalmol. 2009;93(1):21-3. PubMed
- Aerts I, Lumbroso-Le Rouic L, Gauthier-Villars M, et al. Retinoblastoma. Orphanet J Rare Dis. 2006; 25(1):31. PubMed
- Shinohara ET, DeWees T, Perkins SM. Subsequent malignancies and their effect on survival in patients with retinoblastoma. Pediatr Blood Cancer. 2014;61(1):116-9. PubMed
- Rao R, Honavar SG. Retinoblastoma. Indian J Pediatr. 2017; 84:937-44. PubMed
- Skalet AH, Gombos DS, Gallie BL, et al. Screening children at risk for retinoblastoma: Consensus report from the American Association of Ophthalmic Oncologists and Pathologists. Ophthalmology. 2018;125(3):453-8. PubMed
- Scharoun JH, Han JH, Gobin YP. Anesthesia for ophthalmic artery chemosurgery. Anesthesiology. 2017;126(1):165-72. PubMed
- Nghe MC, Godier A, Shaffii A, et al. Prospective analysis of serious cardiorespiratory events in children during ophthalmic artery chemotherapy for retinoblastoma under a deep standardized anesthesia. Paediatr Anaesth. 2018; 28(2):120-6. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.