Copy link
Perioperative Allergies and Anaphylaxis
Last updated: 04/03/2024
Key Points
- Perioperative anaphylaxis (POA) is a life-threatening immediate hypersensitivity reaction that is usually immunoglobulin E (IgE)-mediated. It occurs rarely but may lead to significant morbidity or even mortality.
- Neuromuscular blocking agents (NMBAs) and antibiotics are the main causal agents. Other agents include latex, dyes, antiseptics, colloids, iodinated contrast agents, and sugammadex.
- A tryptase level obtained approximately 1 hour after reaction onset can aid in the diagnosis, but a normal level does not rule out the diagnosis.
- Administration of intravenous epinephrine and the expansion of intravascular volume are key points in the perioperative management of anaphylaxis.
Introduction
- Anesthesiologists use a myriad of drugs during the provision of an anesthetic. Many of these drugs have side effects that are dose-related, and some lead to severe immune-mediated adverse reactions. Anaphylaxis is the most severe immune-mediated reaction.
- It generally occurs upon re-exposure to a specific antigen and requires the release of proinflammatory mediators.
- Signs and symptoms may include all organ systems and present with bronchospasm and cardiovascular collapse in the most severe cases.
- The management of anaphylaxis includes discontinuation of the presumptive drug and anesthetic, aggressive pulmonary and cardiovascular support, and the administration of epinephrine.1
Epidemiology
- The National Audit Project 6 (NAP6) in the United Kingdom estimated the incidence of severe POA at 1 in 10,000.
- POA is less common in the pediatric population, as shown by the NAP6 estimate of severe POA in children at 1 in 37,000.
- There is a female predilection with a female:male ratio of 3:1.2. Possible contributors include the female hormones’ influence on the type 1 helper T-cell/type 2 helper T-cell balance or the propensity for female patients to be sensitized to NMBAs through common consumer products.
- POA carries a higher mortality rate than other causes of anaphylaxis, with estimates ranging from 1.4% to 9%.2
Pathophysiology
- Anaphylaxis is a type I immunoglobulin (Ig)E-mediated hypersensitivity reaction involving mast cells and basophils. On the other hand, contact dermatitis is a type IV T-lymphocyte cell-mediated delayed-type hypersensitivity reaction.1
- Other immune-mediated reactions include type II reactions in which IgG, IgM, and complement mediate cytotoxicity and type III reactions in which immune-complex formation and deposition lead to tissue damage.
- On initial exposure to an antigen in susceptible individuals, IgE is produced and binds to mast cells and basophils. Upon re-exposure, the multimeric antigen cross-links two IgE receptors, inducing the tyrosine phosphorylation of their cytoplasmic immune tyrosine activation motifs by Lyn and Syk tyrosine kinases. This initiates a signal-transduction cascade, which culminates in the increase of intracellular calcium and the release of preformed mediators such as histamine, proteases (tryptases), proteoglycans, and platelet-activating factor. Phospholipid metabolism then leads to the generation of potent inflammatory leukotrienes (LTC4, LTE4, and LTD4) and prostaglandins (PGD2). Histamine, PGD2, and LTC4 are potent vasoactive mediators implicated in vascular permeability changes, flushing, urticaria, angioedema, hypotension, and bronchoconstriction (Figure 1).1
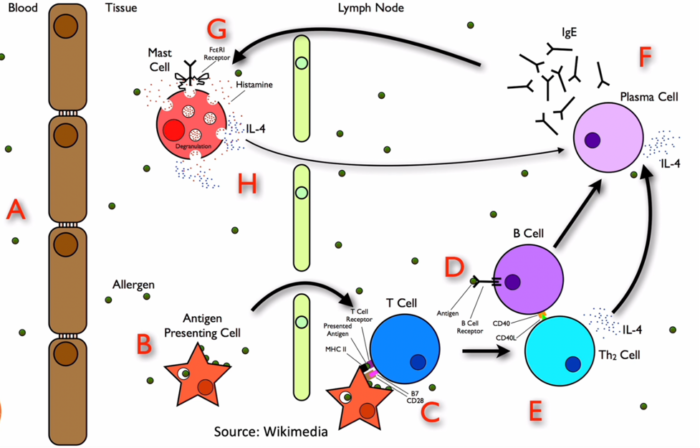
Figure 1. Pathophysiology of anaphylaxis reactions. Source: Wikimedia
- POA is a life-threatening immediate hypersensitivity condition, and the main causal agents are NMBAs and antibiotics. Drugs commonly involved in POA are listed in Table 1.
- Anaphylaxis due to latex can present with delayed onset, often greater than 30-60 min after initial exposure. Risk factors for latex allergy include chronic latex exposure (e.g., health care workers) and a history of atopy. Cross-reactivity between latex and food allergies have been reported (e.g., mango, kiwi, chestnut, avocado, passionfruit, banana).
- Other agents include dyes (patent blue, methylene blue), antiseptics (chlorhexidine, povidone iodine), colloids (gelatins), iodinated contrast agents, and sugammadex.3
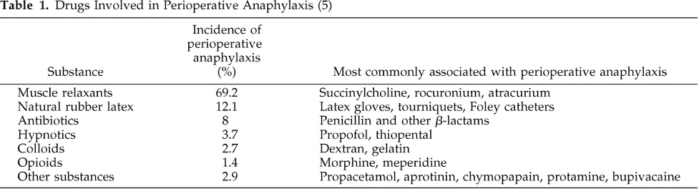
Table 1. Drugs involved in perioperative anaphylaxis. Used with permission from Hepner DL, Castells MC. Anaphylaxis during the perioperative period. Anesth Analg. 2003.1
- Neuromuscular blockers4
- One of the most common culprits for POA
- Europe/Australia (50-70%)>>North America (30%)
- Binds IgE via tertiary or quaternary ammonium structure
- Sensitization via cosmetics, disinfectants, OTC medications
- Non-IgE mediated (Atracurium)-histamine release
- Usually, early onset POA
- Succinylcholine and rocuronium- highest risk
- NAP6-Succinylcholine-highest rate of POA
- Cross-reactivity is common
- Antibiotics4
- Most common culprit in the US-Cefazolin
- >90% of patients are incorrectly labeled as penicillin allergic
- Usually, early in surgery
- Usually, IgE-mediated reaction
- Vancomycin-can cause Redman syndrome
- Latex allergy4
- Natural product found in rubber tree-Hevea brasiliensis
- Used to be ubiquitous in medical equipment
- Allergic contact dermatitis, urticaria, anaphylaxis
- Type 1 IgE-mediated (anaphylaxis) or type 4 cell-mediated hypersensitivity reaction
- 4% in the general pediatric population
- Sugammadex4
- Cyclodextrin compound
- Trigger-cyclodextrin exposure via food additives, cosmetics, etc.
- Sugammadex-rocuronium complex
- Usually, late in surgery
- Chlorhexidine2
- Accounts for 9-10% of cases
- Usually late onset-mucocutaneous reaction
- Often overlooked as a cause
- Dyes2
- Blue dyes: patent blue V, Isosulfan blue, and Methylene blue
- Usually, delayed onset reaction
Signs and Symptoms
- Anaphylaxis is generally an unanticipated severe allergic reaction, often explosive in onset, that can occur perioperatively, especially during a surgical procedure when multiple drugs are administered during the conduction of an anesthetic.1
- Because patients are under drapes and mostly unconscious or sedated, the early cutaneous signs of anaphylaxis are often unrecognized, leaving bronchospasm and cardiovascular collapse as the first recognized signs of anaphylaxis1 (Table 2).
- A clinical grading of anaphylaxis has been proposed by Ring and Messmer.5
- Grade I includes cutaneous–mucous signs (erythema, urticaria with or without angioedema).
- Grade II includes mild cutaneous–mucous features ± hypotension ± tachycardia ± dyspnea ± gastrointestinal disturbances.
- Grade III is life-threatening single or multivisceral signs: cardiovascular collapse, tachycardia or bradycardia, ± arrhythmias ± bronchospasm ± cutaneous-mucus signs ± gastrointestinal disturbances.
- Grade IV is cardiac arrest.
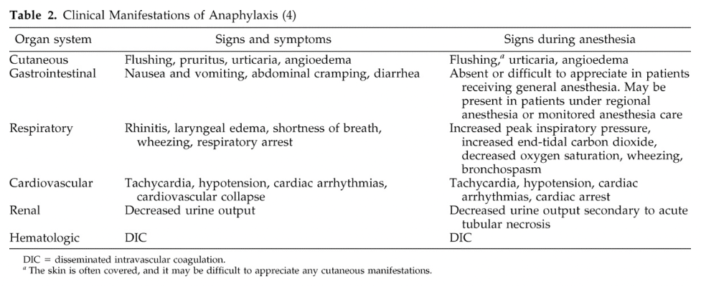
Table 2. Clinical manifestations of anaphylaxis. Used with permission from Hepner DL, Castells MC. Anaphylaxis during the perioperative period. Anesth Analg. 2003.1
- Another potentially confounding sign might be bradycardia, which can be seen in massive hypovolemia, probably as a result of the Bezold–Jarisch reflex. This paradoxical bradycardia occurring during extreme hypovolemia has been reported to occur in as many as approximately 10% of patients with anaphylaxis during anesthesia.6
- Timing of perioperative anaphylaxis2
- Within 30 minutes of induction (95%): antibiotics, NMBAs, hypnotics
- 30 minutes after induction (5%): Latex, sugammadex, disinfectants, blood products
- Differential diagnosis for anaphylaxis under general anesthesia4
- Cutaneous signs
- Histamine release
- Venous obstruction
- Head down position
- C-1 esterase deficiency
- Mastocytosis
- Hypotension
- Hypovolemia
- Peripheral vasodilation by drugs
- Sepsis
- Embolism-air, pulmonary embolism
- Vasovagal
- Cardiogenic shock
- High airway pressures and wheezing
- Circuit malfunction
- Kinked endotracheal tube
- Tension pneumothorax
- Asthma/bronchospasm
- Airway foreign body
- Aspiration
- Cutaneous signs
Diagnosis
- The initial diagnosis of perioperative anaphylaxis relies on the history and physical examination. The retrospective diagnosis is based on serologic and skin tests.
- Serum tryptase is a mast cell protease that is increased in cases of anaphylaxis, signaling an immune-mediated mechanism. An increase in human α and β tryptase, the predominant mast cell proteases, can be measured in serum and plasma 30 min after the first signs of anaphylaxis and correlate with the presence of hypotension.
- An increase of serum tryptase does not differentiate an anaphylactic from an anaphylactoid reaction. Similarly, an absence of serum tryptase does not eliminate an anaphylactic reaction, because there have been reports of anaphylaxis with positive tests for IgE antibodies in the setting of an absence of serum tryptase.1
- Simple algorithm for the diagnosis of perioperative anaphylaxis (Figure 2). The initial diagnosis of perioperative anaphylaxis relies on the history and physical examination. Whereas in vitro tests are used to confirm the diagnosis (serum tryptase) and to identify the offending drug or substance (radioallergosorbent test; RAST), in vivo tests (e.g., skin test) are used to identify the offending drug or substance with more sensitivity and specificity. IgE = immunoglobulin E.1
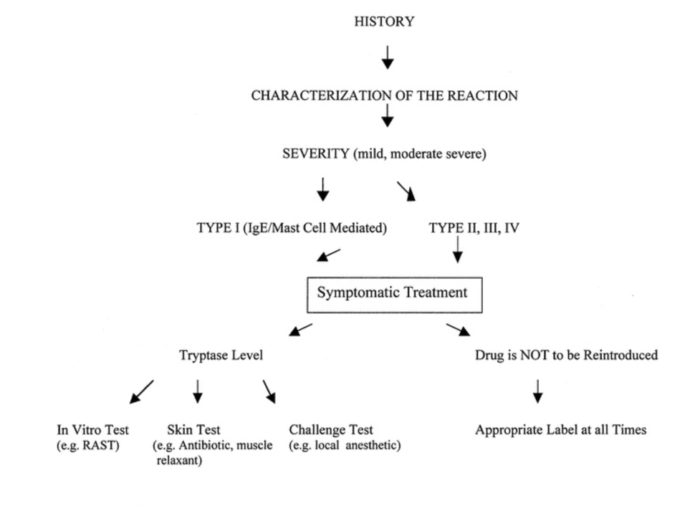
Figure 2. Simple algorithm for the diagnosis of perioperative anaphylaxis. Used with permission from Hepner DL, Castells MC. Anaphylaxis during the perioperative period. Anesth Analg. 2003.1
Management
- The treatment of anaphylaxis includes the following (Table 3).6
- Withdraw the suspected culprit drug
- Immediately discontinue anesthetic drugs when the anaphylactic event occurs during induction
- Administer 100% oxygen
- Support the airway
- Administer epinephrine early, especially in case of grade III or IV reactions
- Call for help, especially for grade III and IV reactions
- Place the patient in the Trendelenburg position
- Abbreviate the surgical procedure, if possible
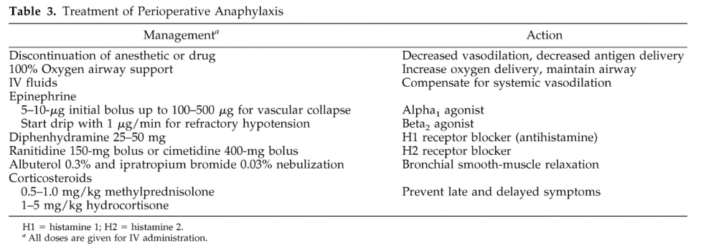
Table 3. Treatment of perioperative anaphylaxis. Used with permission from Hepner DL, Castells MC. Anaphylaxis during the perioperative period. Anesth Analg. 2003.1
- Anaphylactic Shock Refractory to Catecholamines: Alternative Therapy
- Epinephrine sometimes fails to restore the profound disturbances of cardiovascular homeostasis. Norepinephrine, metaraminol, or glucagon for patients receiving β-blocker therapy is recommended in this clinical setting.6
- Because desensitization of adrenergic receptors might be one of the contributing factors of catecholamine failure occurring during anaphylaxis, arginine vasopressin (AVP) may be an alternative through its vasoconstrictive effects mediated by nonadrenergic vascular AVP V1receptors.
- Another factor contributing to catecholamine failure may involve nitric oxide (which seems to play a pivotal role during anaphylaxis) because increased nitric oxide synthesis contributes to the hypotension and resistance to vasopressors during vasodilatory shock.6
References
- Hepner DL, Castells MC. Anaphylaxis during the perioperative period. Anesth Analg. 2003;97(5):1381-95. PubMed
- Mitchell M. Pitlick, Gerald W, Volcheck. Perioperative Anaphylaxis. Immunol Allergy Clin N Am 42 (2022) 145–159. PubMed
- Dewachter P, Savic L. Perioperative anaphylaxis: pathophysiology, clinical presentation and management. BJA Educ. 2019;19(10):313-20. PubMed
- Stepanovic B, Sommerfield D, Lucas M, von Ungern-Sternberg BS. An update on allergy and anaphylaxis in pediatric anesthesia. Paediatr Anaesth. 2019;29(9):892-900. PubMed
- Ring J, Messmer K. Incidence and severity of anaphylactoid reactions to colloid volume substitutes. Lancet. 1977;1(8009):466-469. PubMed
- Dewachter P, Mouton-Faivre C, Emala CW. Anaphylaxis and anesthesia: controversies and new insights. Anesthesiology. 2009;111(5):1141-1150. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.