Copy link
Pediatric Tracheal Extubation
Last updated: 01/25/2023
Key Points
- Tracheal extubation is a critical step in the emergence from general anesthesia.
- Planning and preparation before extubation should include a detailed evaluation of patient comorbidities and airway-related risk factors.
- Other logistics, such as the timing, location, personnel, and equipment, must be optimized prior to extubation.
Introduction
Tracheal extubation is a critical step in the emergence from general anesthesia, yet this topic is often neglected in the literature and generates less interest than intubation.1 The APRICOT study, a prospective multicenter observational study that analyzed more than 31,000 anesthetics in children across 261 hospitals in Europe, reported more respiratory complications such as laryngospasm, bronchospasm, aspiration, and stridor during extubation.2 As an analogy, more aviation accidents occur during landing than takeoff.
Approach to Tracheal Extubation
Several frameworks have been proposed for planning and executing tracheal extubation. The Difficult Airway Society’s guidelines for tracheal extubation in adults can be modified for use in children.3 More recently, Weatherall et al. have proposed a R2D2 framework for extubation in children.4
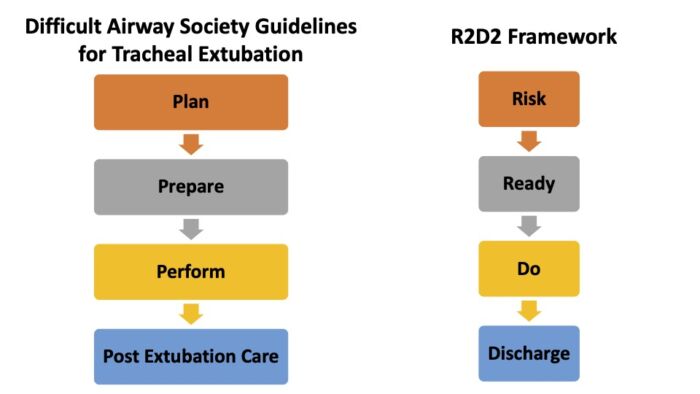
Figure 1. Examples of approaches to pediatric tracheal extubation.
Step 1: Planning and Risk Assessment
Planning and assessing the risk of a failed extubation is a critical first step for success. The presence of any airway-related risk factors and patient comorbidities must be evaluated.
Pre-Existing Airway-Related Risk Factors3,4
- Difficult bag-mask ventilation requiring:
- airway adjuncts, significant positive pressure, two-person technique, etc.
- Difficult intubation
- Grade 3 or 4 Cormack and Lehane view on direct laryngoscopy
- Video laryngoscopy view with <50% of vocal cords
- Multiple (3+) intubation attempts
- Prior failed intubation
- Obesity/obstructive sleep apnea
- Patient at high risk for aspiration
New Airway-Related Risk Factors3,4
- Perioperative airway deterioration
- Glottic or subglottic edema
- Massive hemorrhage
- Dependent edema from positioning
- Aggressive fluid resuscitation
- Restricted airway access
- Halo fixation
- Cervical spine fixation/collar
- Mandibular wiring
Patient Comorbidities3,4
- Impaired respiratory function
- Cardiovascular instability
- Neurological impairment
- Hypo/hyperthermia
- Abnormal clotting
- Abnormal acid-base balance, electrolytes
Awake vs. Deep Extubation
If the patient has any pre-existing or new airway-related risk factors or patient comorbidities listed above, awake extubation should be performed. There are certain conditions when deep extubation is preferred:1
- children with reactive airway disease or asthma to avoid bronchospasm;
- following certain surgeries where coughing and bucking on the endotracheal tube (ETT) during extubation can be detrimental, such as middle ear or open eye surgeries.
Step 2: Preparation and Getting Ready for Extubation
The next step is to optimize patient factors and logistics in preparation for extubation.
Return to Spontaneous Ventilation
- The transition from controlled to spontaneous ventilation at the end of the anesthetic depends on several factors such as the depth of anesthesia, dose of opioid administered, residual neuromuscular blockade, patient’s age, temperature, etc.1
- Often pressure support ventilation with rescue breaths or gentle hand ventilation is performed during this transition to avoid atelectasis.
- Before extubation, it is important to ensure that the patient is breathing spontaneously, ideally with tidal volumes more than 5 mL/kg. This can be hard to assess in small neonates and infants, and chest movement can be used as a surrogate marker.1
- It is important to recognize that the diaphragm and intercostal muscles recover before the upper airway muscles, such as the genioglossus. Even in the absence of muscle relaxation, the upper airway dilating muscles are more sensitive to the action of volatile anesthetics than the diaphragm and intercostal muscles. Careful observation of the chest and abdominal movements is thus important to avoid respiratory problems after extubation.1
Reversal of Neuromuscular Blockade
- If a nondepolarizing muscle relaxant (NDMR) was used during the anesthetic, adequate recovery of neuromuscular function must be verified by measuring a train-of-four (TOF) ratio greater than 0.9. Either neostigmine and glycopyrrolate or sugammadex must be administered if there is inadequate recovery. It is important to recognize that even a TOF >0.9 doesn’t guarantee a full recovery of muscle function, especially the upper airway muscles. Regardless, neuromuscular monitoring must be performed routinely in children if an NDMR was used.
Optimizing Other Patient Factors
- Hemodynamic stability
- Adequate analgesia
- Optimal temperature
- Normal acid-base and electrolytes
- Normal coagulation status
Location and Skilled Help
The decision to extubate in the operating room (OR) or postanesthesia care unit (PACU) is a matter of debate and depends on local practices.1 Proponents of extubating in the PACU claim decreased turnover times between cases. If the patient is extubated in the PACU, there must be experienced personnel for the early recognition of complications and the immediate availability of an anesthesiologist and airway equipment. If the patient has any of the airway risk factors mentioned previously, the patient must be extubated in the OR. For ICU patients, a decision must be made in collaboration with the intensivist, anesthesiologist, and otolaryngologist.
Reintubation Plan
A comprehensive reintubation plan must be in place in patients with difficult airways or at high-risk for extubation failure.
Equipment
For routine extubation, the following drugs and equipment should be readily available.4
- Face mask and breathing circuit
- Working intravenous access
- Suction
- Airway adjuncts (oral/nasal airways)
- Emergency drugs: succinylcholine, atropine
For patients with difficult airways or at high-risk for extubation failure, consider the following:4
- Drugs and equipment for reintubation: induction agent, ETTs, supraglottic airways, etc.
- Difficult airway equipment: choice depends on availability and clinician preference
- Equipment for respiratory support: noninvasive ventilation, humidified high-flow nasal cannula, nebulizer for racemic epinephrine, etc.4
Bypassing Upper Airway Obstruction
In patients with severe OSA, microretrognathia, or following surgeries where tongue-swelling is expected (prone positioning), a well-lubricated nasopharyngeal airway may be placed to bypass the upper airway obstruction.1
Reducing Coughing on Emergence
Several medications can be administered to reduce coughing on emergence from general anesthesia. A recent systematic review and network meta-analysis in adults identified the administration of dexmedetomidine as the most effective, followed in order by remifentanil, fentanyl, and lidocaine via intracuff, tracheal/topical and intravenous routes.5 The benefits of cough suppression must be weighed against the risk of sedation and respiratory depression.3
Positioning for Extubation
While there is no evidence to support a universal patient position for extubation, using a semirecumbent or reverse Trendelenburg position offers a mechanical advantage in obese children. Some clinicians use the left-lateral head-down positions for patients at risk for aspiration.3
Step 3: Perform Extubation
The steps in awake and deep extubation are listed in Figure 2. Some considerations include:
Inspired Oxygen Concentration (FiO2)
While most textbooks and references cite using 100% O2 just prior to extubation, some authors recommend using a FiO2 of 0.8 or lower to reduce absorption atelectasis and the production of reactive oxygen metabolites.1 The clinical significance of using a high FiO2 is yet to be determined.3
Timing of Extubation
Just prior to extubation, a common practice is to apply positive pressure, deflate the cuff, and remove the ETT. Some authors recommend extubating at the end of inspiration.1
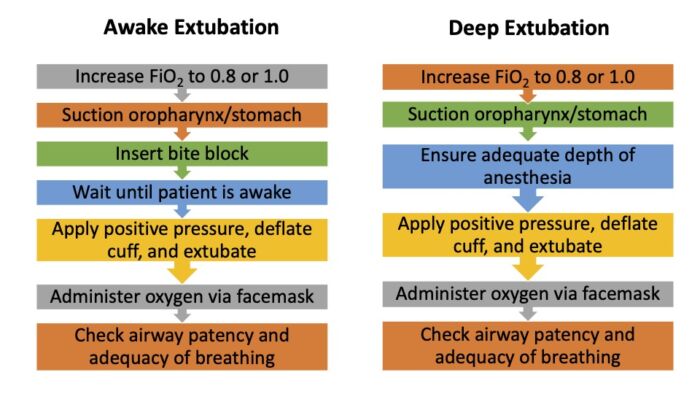
Figure 2. Steps in awake and deep extubation.
Probability of Successful Extubation
Templeton et al. conducted a prospective, observational study in 600 children (0-7 years of age) undergoing awake tracheal extubation after a volatile anesthetic and identified 5 criteria that were associated with successful extubation.6
- Breathing spontaneously with tidal volumes > 5 mL/kg
- Conjugate gaze
- Facial grimace
- Eye-opening
- Purposeful movement
Additionally, the higher the number of criteria present indicated the greater likelihood of extubation success (Table 1).
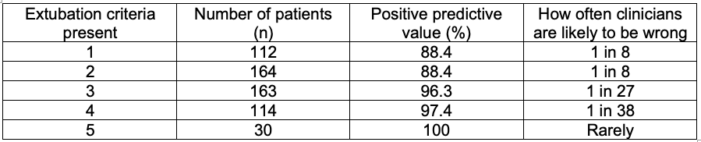
Table 1. Predictive value of extubation criteria for awake extubation in children. Adapted from Templeton TW, et al. Extubation of pediatric patients following general anesthesia. Anesthesiology News Airway Management. 2020.
Extubation to Noninvasive Ventilation
Extubation to bilevel positive airway pressure (BiPAP) or continuous positive airway pressure (CPAP) may be considered for the following patients.
- Chronic respiratory failure
- Neuromuscular disease
- Advanced cystic fibrosis
Contraindications include
- Unstable hemodynamics
- Aspiration risk
- Altered mental status
- Facial trauma
Step 4: Postextubation Care
Following extubation, pay attention to the following:1,3,4
- Administration of oxygen during transport to PACU and continued thereafter
- Continuous monitoring of the patient in the PACU
- Detailed hand-off to nursing staff in the PACU
- Early recognition and prompt treatment of complications such as upper airway obstruction, laryngospasm, bronchospasm, central apnea, vomiting, aspiration, etc.
- Assessment of the ongoing need for analgesia and/or sedation
- Appropriate documentation of the extubation process and any issues noted
References
- Veyckemans F. Tracheal extubation in children: Planning, technique, and complications. Pediatr Anesth. 2020; 30:331–8. PubMed
- Engelhardt T, Virag K, Veyckemans F, et al. Airway management in paediatric anaesthesia in Europe-insights from APRICOT (Anaesthesia PRactice In Children Observational Trial): a prospective multicenter observational study in 261 hospitals in Europe. Br J Anaesth. 2018; 121:66-75. PubMed
- Difficult Airway Society Extubation Guidelines Group; Popat M, Mitchell V, Dravid R, et al. Difficult Airway Society guidelines for the management of tracheal extubation. Anaesthesia. 2012; 67(3): 318-40. PubMed
- Weatherall AD, Burton RD, Cooper MG, et al. Developing an extubation strategy for the difficult pediatric airway-Who, when, why, where, and how? Pediatr Anesth. 2022; 32:592–9. PubMed
- Tung A, Fergusson NA, Ng N, et al. Medications to reduce emergence coughing after general anesthesia with tracheal intubation: a systematic review and network meta-analysis. Br J Anaesth. 2020;124(4): 480-95. PubMed
- Templeton TW, Goenaga-Diaz EJ, Downard MG, et al. Assessment of common criteria for awake extubation in infants and young children. Anesthesiology. 2019; 131(4): 801-8. PubMed
Other References
- Chatterjee D. Pediatric tracheal extubation. OpenAnesthesia. Published October 2021. Accessed January 25, 2023. Link
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.