Copy link
Pediatric Obstructive Sleep Apnea
Last updated: 03/08/2023
Key Points
- Most pediatric patients with obstructive sleep apnea syndrome (OSAS) will not have undergone formal testing nor have a formal diagnosis of OSAS; however, those with severe OSAS may have a sensitivity to opioid medications.
- While multiple factors predispose to OSAS, adenotonsillar hypertrophy and obesity are the major risk factors in otherwise healthy pediatric patients.
- Patients with OSAS are at an increased risk of respiratory complications and upper airway obstruction after anesthesia. Anesthetic management should aim to reduce factors that contribute to respiratory depression and airway collapse, optimize airway management, and have contingency plans should the patient have airway obstruction postoperatively.
Introduction
- OSAS is a sleep disorder characterized by partial or complete nocturnal upper airway obstruction affecting approximately 1-6% of all children and up to 59% of obese children.1
- Untreated OSAS is associated with effects on multiple systems, including behavioral disorders, poor school performance, enuresis, metabolic abnormalities, cardiovascular disease, including pulmonary hypertension, and pulmonary complications.
Risk Factors
- Pediatric OSAS is primarily caused by adenotonsillar hypertrophy and obesity, but craniofacial abnormalities and neuromuscular diseases can also cause OSAS (Table 1).1,2
- Adenotonsillar hypertrophy is more prominent in ages 2-8 years. Size is influenced by genetics, infection, and inflammation; however, studies have not shown a linear relationship between tonsil size and degree of airway obstruction.
- Obesity as a risk factor affects all ages, but more in older children and teens.
- Other predisposing factors include:
- history of prematurity and multiple gestation;
- history of low birth weight;
- craniofacial anomalies (e.g., retrognathia, micrognathia, midface hypoplasia, Pierre Robin sequence, achondroplasia);
- orthodontic problems (e.g., high narrow hard palate, overlapping incisors, crossbite); and
- family history of OSA.2
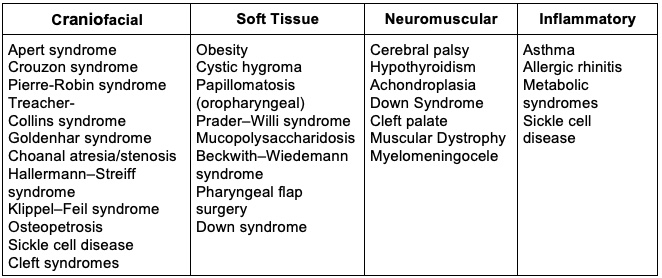
Table 1. Congenital and medical conditions associated with obstructive sleep apnea1
Pathophysiology
- The upper airway is subject to collapse based on upstream and downstream pressure differences. Upstream and downstream pressures must be greater than the critical pressure (known as PCRIT).1
Anatomic Obstruction
- Anatomic obstruction can be due to airway narrowing from adenotonsillar hypertrophy or decreased space in the posterior oropharynx due to craniofacial abnormalities. Airway obstruction in pediatric OSAS patients under anesthesia is more likely to occur at the level of the tonsils and adenoids compared to the soft palate in adults.
Neuromechanical Dysfunction
- OSAS patients have altered neuromuscular responses to changes in airway pressures. Unlike in adults, children can maintain their airway opening and often do not have full collapse, leading to a low-flow breathing pattern that characteristically leads to hypercarbia. Children with OSAS often have an increased arousal threshold to carbon dioxide.
Neuromuscular Dysfunction
- Hypotonia in diseases such as Trisomy 21 predisposes patients to airway collapse due to neuromuscular dysfunction and airway obstruction from macroglossia. Obesity also predisposes to airway collapse due to increased pressure from excess tissues around the airway.
Inflammation
- Chronic inflammatory disorders such as sinusitis, allergic rhinitis, and asthma as well as environmental smoke exposure are all associated with elevated inflammatory markers and airway collapse in OSAS.1
Genetic
- Genetic predisposition has been demonstrated in family cohort studies. African American children are at 3.5 times higher risk for OSAS than other populations.1
Diagnosis
- Nocturnal polysomnography (PSG) is the gold standard for diagnosing OSAS, but fewer than 10% of children with sleep disordered breathing present for anesthesia and surgery having had a PSG.3
- Surgeons will often diagnose patients based on physical airway exam and descriptions by parents of breathing patterns during sleep (Table 2).
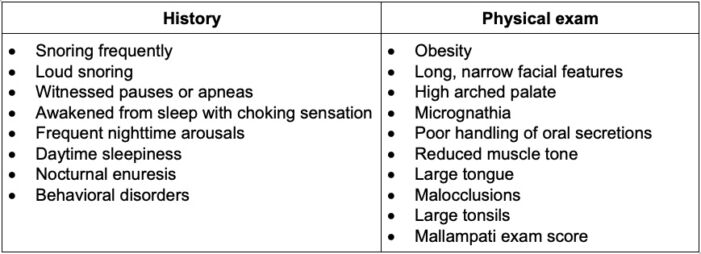
Table 2. Focused history and physical exam findings that may be suggestive of OSAS1
Polysomnography
- PSG measures the apnea/hypopnea index (AHI), an average of apnea and hypopnea episodes per hour of sleep.
- A score greater than or equal to 1 is consistent with mild OSA, where severe OSAS is diagnosed when AHI is more than 10.

Table 3. Definitions of apnea and hypopnea1

Table 4. Apnea Hypopnea Index definition criteria for severity of OSAS1
Management
- Surgery
- Adenotonsillectomy is the most common surgical treatment for children with OSAS.
- Complete adenotonsillectomy
- Partial or subcapsular tonsillectomy
- Higher risk of tonsillar regrowth and recurrence of OSAS.
- Lower risk of posttonsillar hemorrhage and pain
- Pharyngeal surgery, craniofacial surgery, supraglottoplasty
- Nasal continuous positive airway pressure or bi-level positive airway pressure
- Inhaled or nasal steroids
- Orthodontic interventions for those with maxillofacial abnormalities
- Weight loss1
Anesthetic Considerations
Preoperative Evaluation
- Anesthesiologists must maintain a high index of suspicion for OSAS in patients with the above history and exam findings, given that not all children with OSAS will have a formal diagnosis.
- For all patients with suspected OSAS (and/or those undergoing adenotonsillectomy), a thorough preoperative evaluation should be performed focusing on:
- PSG (if available): assessing peak etCO2, AHI score, oxygen saturation nadir, etc.
- Specialist documentation (ENT and pulmonology evaluation)
- Thorough history and physical exam1,2
- Consider an echocardiogram to assess right heart function, signs of pulmonary hypertension, left heart function, and systemic hypertension if cardiac symptoms are present.
Intraoperative and Postoperative Management
- Pediatric patients with OSAS are at higher risk of perioperative complications compared to those without OSAS.1-2,4 Anesthetic management should focus on minimizing factors predisposing to airway collapse or worsening obstruction. Furthermore, clinicians must be prepared to manage airway obstruction at any point perioperatively.
- Clinicians should be prepared to manage airway obstruction via oropharyngeal airway, nasal trumpet, supraglottic airway or bag-mask ventilation during induction and emergence from anesthesia.
- Hypoxemia and hypercarbia should be avoided as OSAS patients are more prone to these physiologic imbalances and may worsen OSAS symptoms.
- Pain management should focus on optimizing nonopioid analgesics along with the judicious administration of opioids.
- Clinicians should consider use of nonopioid analgesics such as acetaminophen, NSAIDs, ketamine, and dexmedetomidine.1,5
- Clinicians should consider use of local anesthetic infiltration, regional anesthesia and neuraxial anesthesia for patients undergoing nonairway surgeries.
- Judicious use of opioids in the setting of altered response should be considered.
- Patients with pediatric OSAS are more sensitive to the respiratory depressant effects of opioids. This is thought to be due to the upregulation of opioid receptors during hypoxic episodes.7
- Studies show increased morphine metabolism in obese pediatric patients with OSAS compared to those with OSAS alone; however, due to morphine’s hydrophilic properties, more drug may stay intravascular and affect central respiratory centers leading to respiratory depression in obese patients.5
- Patients with OSAS undergoing adenotonsillectomy should receive dexamethasone (0.3 – 0.5 mg/kg; max dose of 24 mg) as it has been shown to reduce airway swelling and opioid use.8
- Pharmacology: Clinicians should consider avoiding overly sedating medications and dose hydrophilic medications based on ideal body weight in obese individuals.
- Awake extubation in patients with severe OSAS should be considered.
- Prolonged monitoring postoperatively or ICU admission should be considered depending on the severity of OSAS, age, comorbidities, and type of surgery performed.6
References
- Dalesio NM, Hayward DM, Schwengel DA. Anesthesia for pediatric otorhinolaryngologic surgery. In: Smith's anesthesia for infants and children, 9th ed. Elsevier Health Sciences, 2016: 817-42.
- Paruthi S. Evaluation of suspected obstructive sleep apnea in children. In: Post T, ed. UpToDate. 2022. Accessed August 23, 2022.
- Mitchell RB, Pereira KD, Friedman NR. Sleep-disordered breathing in children: Survey of current Practice. Laryngoscope. 2006; 116:956–8. PubMed
- Schwengel DA, Sterni LM, Tunkel DE, Heitmiller ES. Perioperative management of children with obstructive sleep apnea. Anesth Analg. 2009;109(1):60-75. PubMed
- Dalesio NM, Lee CKK, Hendrix CW, et al. Effects of Obstructive Sleep Apnea and Obesity on Morphine Pharmacokinetics in Children. Anesth Analg. 2020;131(3):876-84. PubMed
- Smith DF, Spiceland CP, Ishman SL, et al. Admission Criteria for Children with Obstructive Sleep Apnea After Adenotonsillectomy: Considerations for Cost. J Clin Sleep Med. 2017;13(12):1463-72. PubMed
- Brown KA, Laferriere A, Moss IR. Recurrent hypoxemia in young children with obstructive sleep apnea is associated with reduced opioid requirement for analgesia. Anesthesiology. 2004;100(4):806-10. PubMed
- Mitchell RB, Archer SM, Ishman SL, et al. Clinical practice guideline: tonsillectomy in children (Update). Otolaryngol Head Neck Surg 2019;160: S1–42. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.