Copy link
Organ Donation: Donation After Brain Death and Donation After Cardiac Death
Last updated: 05/24/2023
Key Points
- Donation after brain death (DBD) involves donation of organs after the patient meets criteria for death by neurological criteria. Donation after cardiac death (DCD) involves donation of organs after irreversible cessation of circulatory and respiratory function.
- For DCD donors, warm ischemic time in addition to cold ischemic time determines organ viability.
- Allowable ischemic times are based upon the organ being donated.
Donation After Brain Death vs Donation After Cardiac Death
Donation After Brain Death (DBD)
- Death is confirmed via neurological criteria.
- Brain death diagnosis requires the presence of 3 conditions: persistent coma, absence of brainstem reflexes, and a lack of the ability to breathe independently.
Donation After Cardiac Death (DCD)
- Death is confirmed via cardiorespiratory criteria (cardiac arrest) normally after withdrawal of support.
- Controlled donation: planned withdrawal of support (most cases)
- Uncontrolled donation: cardiac arrest is unexpected (few)
- Transplant team members cannot be involved in decisions related to patient prognosis, withdrawal of ventilatory or organ-perfusion support or determination of death as this represents a conflict of interest.
DCD Donor Characteristics
- Organs are medically suitable for donation.
- Brain death criteria is not met, although a potential DCD donor may progress to DBD donor.
- Patient has suffered a catastrophic brain injury or other illness such as end-stage musculoskeletal disease, pulmonary disease, or high spinal cord injury.
- There is no expectation of meaningful survival (per treating physician).
- Request for withdrawal of ventilatory and organ-perfusion support is made by the patient’s legal decision-maker and/or by properly executed prior first-party declarations.
- Informed consent is obtained from the patient’s legal decision maker(s) (obtained after decision to withdraw support).
- The possibility that the patient may not die or may not provide transplanted organs must be communicated to the consenter.
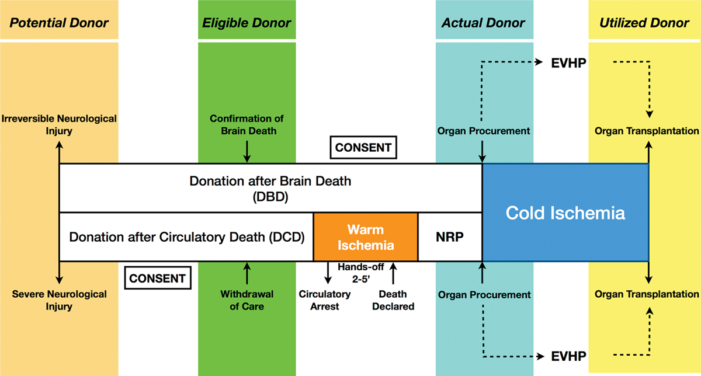
Figure 1. Organ Donation Protocols. Comparison of donation pathways for DBD vs DCD. Abbreviations: EVHP, ex vivo heart perfusion; NRP, normothermic regional perfusion. Reproduced with permission from Beuth J, et al. New strategies to expand and optimize heart donor pool: Ex-vivo heart perfusion and donation after circulatory death: A review of current research and future Trends. Anesth Analg. 2019;128(3):406-13.
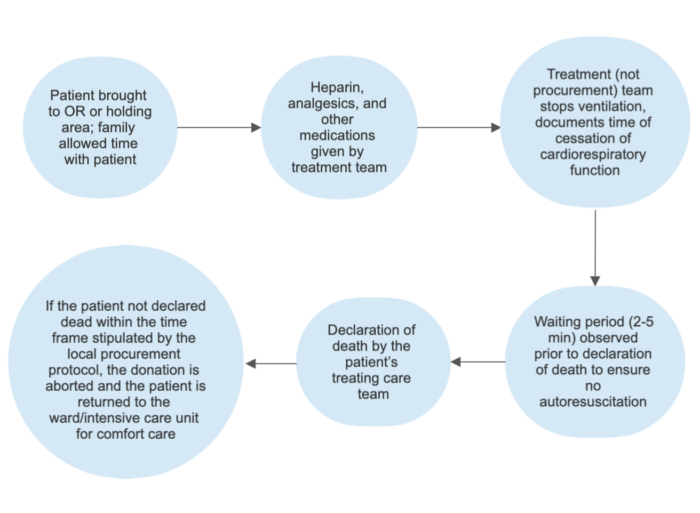
Figure 2. Schematic of DCD withdrawal of support protocol
Outcomes
- For most organs when compared with DBD, DCD provides a lower yield of transplantable organs, decreased patient and graft survival rates, and increased delayed graft function. Therefore, it is preferred that organ donation be pursued according to DBD rather than DCD protocols, if possible.2
- However, DCD can provide an expansion of the organ donor pool if DBD is not feasible, such as when there is catastrophic injury but brain death is not imminent and there is a request for withdrawal of ventilatory and organ-perfusion support or completion of a brain-death protocol with a seemingly brain-dead or soon-to-be brain-dead potential organ donor is precluded because of severe hemodynamic instability or because the family/legal decision-makers will not wait for completion of a brain-death protocol.
Donation After Brain Death- Ischemic Times
- In DBD patients, organ viability is determined based upon cold ischemic items or the time from aortic cross-clamping to reperfusion. The following cold ischemic times are considered optimal for DBD organ donation. Longer ischemic times are highly correlated with increased rate of primary nonfunction and associated with delayed graft function.
- Liver: 8-10 hours
- Kidneys: 24 hours
- Pancreas: 12-18 hours
- Heart: 4 hours
- Lungs 4-6: hours
- Intestine: 6-12 hours
Donation After Cardiac Death: Ischemic Times
Warm Ischemic Time
- If the donor is a DCD donor, the organ has both cold and warm ischemic time (as above). Warm ischemic time refers to the amount of time that an organ remains at body temperature after its blood supply has been stopped or reduced or practically speaking, warm ischemic time is the time from agonal phase onset or drop in blood pressure below a threshold to the time when aortic clamping occurs and core cooling is initiated.2
- Efforts should be made to minimize both warm and cold ischemia times during DCD organ procurement and transplantation.2 Transplantation beyond the following time frames may be associated with increased complications.
Liver
- True warm ischemia time (SBP less than 50 mmHg or SpO2 less than 70 %) greater than 20–30 min
- Total warm ischemia time (interval between discontinuation of mechanical ventilation and initiation of perfusion) greater than 30–45 min
- Cold ischemia time greater than 8–10 hours
Kidneys/Pancreas
- Total warm ischemia time greater than 45–60 min
- Cold ischemia time greater than 24 h for kidneys and greater than 18 h for pancreas
Lungs
- 60 minutes (from onset of functional warm ischemia to mechanical re-inflation of lungs)
- Cold ischemia time greater than 4-6 hours
References
- Beuth J, Falter F, Ribeiro RVP, et al. New strategies to expand and optimize heart donor pool: Ex vivo Heart perfusion and donation after circulatory death: A review of current research and future trends. Anesth Analg. 2019;128(3):406-13. PubMed
- Reich DJ, Mulligan DC, Abt PL, et al. ASTS recommended practice guidelines for controlled donation after cardiac death organ procurement and transplantation. Am J Transplant. 2009; 9(9):2004-11. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.