Copy link
Methyl Methacrylate Complications and Bone Cement Implantation Syndrome
Last updated: 05/09/2023
Key Points
- Methyl methacrylate (MMA) is a volatile and highly flammable monomer commonly used in the medical setting as a cementing agent and adhesive when formed to create its polymer polymethyl methacrylate (PMMA).
- Procedural uses include bonding of orthopedic prosthetics, dental procedures for the creation of dentures, crowns, and bridges, and craniofacial reconstruction.
- Complications that can occur in a procedural setting for the patient include hypotension, intravascular embolization, local dermal irritation, and the potential for hepatotoxicity.
- Complications that can occur to the person handling the MMA include local neurologic effects including numbness, pain, finger blanching, and acute respiratory irritation.
- Current literature does not demonstrate any exposure risk during pregnancy or to a breast-feeding infant.
Introduction
- MMA is a monomer primarily used for the creation of acrylic resin and plastics.1 The monomer form appears as a colorless liquid commonly combined with an inhibitor to prevent the initiation of polymerization.
- In its most common form, MMA is used to create, via polymerization, PMMA.
- MMA is a volatile and highly flammable compound.1
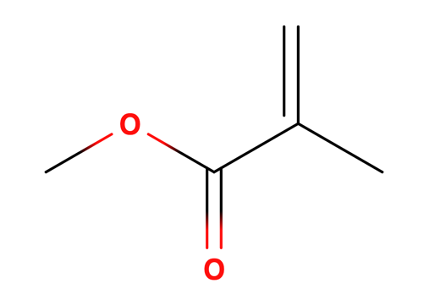
Figure 1. Chemical structure of methyl methacrylate (CH2=C(CH3)COOCH3).
Medical Uses
- In the surgical setting, MMA is most commonly used in orthopedics for cementing joint replacements.
- It is also used as part of the Masquelet technique in orthopedic surgery for lower extremity injuries caused by osteomyelitis.
- In vertebroplasty, it is used via percutaneous injection to restore vertebral body height, reduce pain, and stabilize fractures.
- In dental practice, it is used for the creation of dentures, temporary crowns, and bridges as well as for craniofacial reconstruction.
Complications
- Severe hypercapnia and pulmonary venous embolism from PMMA monomer and polymer are a described but rare complication of vertebroplasty. Clinical findings of embolization include respiratory distress, coughing, hypotension, and decreased end-tidal CO2.2
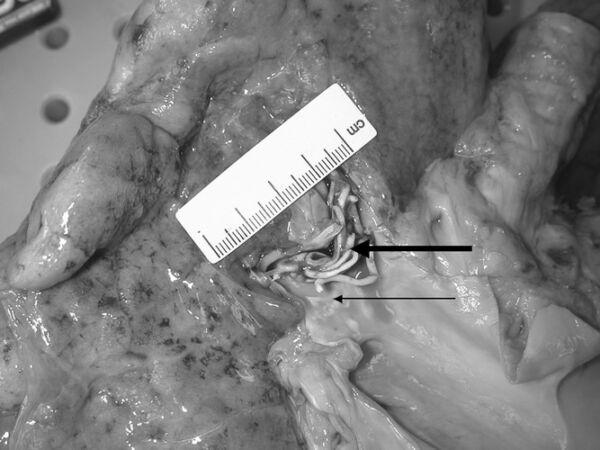
Figure 2. Methyl methacrylate polymer vascular casts embolized and lodged in the right upper and middle lobe arteries at autopsy. Used with permission from Stricker K, et al. Severe hypercapnia due to pulmonary embolism of polymethylmethacrylate during vertebroplasty. Anesth Analg. 2004.2
- The integrity of latex gloves may be compromised when using MMA. Adverse effects associated with direct contact include numbness, pain, and whitening of fingers.3
- Hepatoxicity associated with vessel necrosis and subcapsular hemorrhage has been described. In-vivo studies have demonstrated diffuse microvesicular hepatic steatosis associated with monomer vapor inhalation.4
- Decrease in systolic blood pressure following cement insertion for femoral head replacement with a significant risk of intraoperative hypotension is another complication.
- Acute respiratory irritation from inhalation of MMA has been noted. Long-term effects related to pulmonary function have not been adequately studied.5
- Studies have not found a significant correlation between MMA usage and respiratory irritation with subsequent allergic reactions or asthma.
- Bone cement implantation syndrome is also a possible complication (described below).
Bone Cement Implantation Syndrome (BCIS)
- BCIS is characterized by clinical findings ranging from hypoxia, arrhythmias, and hypotension to cardiac arrest.6
- It is caused by the release of the MMA monomer into the circulation directly following cementation.
- The pathophysiology of hypotension and cardiac arrest is poorly understood with some studies suggesting a secondary cause of right ventricular failure from increased pulmonary artery pressure.6
- The risk is increased in patients with comorbid conditions such as hypertension and obesity as well as with patients who have had a previously uninstrumented femoral canal.6
- A grading of BCIS has been proposed based on the severity of symptoms.6,7
- Grade 1: moderate hypoxia (SpO2 less than 94%) or a decrease in systolic arterial pressure more than 20%
- Grade 2: severe hypoxia (SpO2 less than 88%) or a decrease in systolic arterial pressure more than 40%, or an unexpected loss of consciousness
- Grade 3: cardiovascular collapse requiring resuscitation
- Though there is no clear evidence regarding the anesthetic technique for preventing BCIS, one animal study demonstrated that the use of volatile anesthetics generates greater hemodynamic instability with similar embolic loads.6,7
- Further techniques include increased inspired oxygen concentration at the time of cement insertion.
- Patients at high risk of BCIS should be monitored perioperatively with invasive arterial pressure and central venous catheter.
- The treatment of BCIS is primarily supportive care in line with the treatment of acute right ventricular failure, and includes 100% oxygen, intravenous fluids, inotropes, and pulmonary vasodilators.6
- One study demonstrated that the use of bone cement during total knee arthroplasty when using MMA or PMMA led to a transient astroglial injury when monitoring serum levels of S-100B released from glial and Schwann cells compared to controls. However, there were no recorded neurologic abnormalities in the postoperative period compared to controls.8
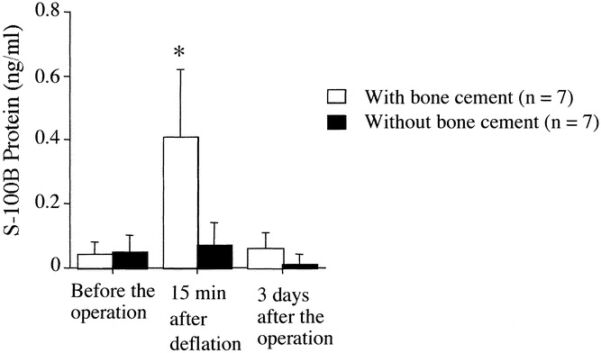
Figure 3. Perioperative serum levels of S-100B protein undergoing bone surgery utilizing cement containing MMA compared to control. Used with permission from Kinoshita H, et al. The use of bone cement induces an increase in serum astroglial S-100B protein in patients undergoing total knee arthroplasty. Anesth Analg. 2003.8
Prevention of Complications
- Appropriate personal protective equipment, including gloves, safety glasses, and a properly fitted N95 mask should be worn.
- Mixing of MMA with the polymerizing agent in a well-ventilated area, preferably under suction if available, has been described to decrease the amount of monomer available in the sample compared to nonventilated mixed samples.
- Correct mixing ratios based on manufacturer’s protocols should be used.
- Mixing should be performed at a slow and stable pace to avoid intense exothermic reactions and excess vaporization.
- Waste material should be disposed properly.
- Contact of material on skin or areas on the patient that do not require the material should be avoided.
- Fluoroscopy in vertebroplasty procedures should be utilized to ensure proper location of injection of the polymer and avoid systemic toxicity.
References
- National Center for Biotechnology Information. PubChem Compound Summary for CID 6658, Methyl methacrylate. Accessed Feb. 5, 2023. Link
- Stricker K, Orler R, Yen K, et al. Severe hypercapnia due to pulmonary embolism of polymethylmethacrylate during vertebroplasty. Anesth Analg. 2004;98(4):1184-6. PubMed
- Leggat PA, Smith DR, Kedjarune U. Surgical applications of methyl methacrylate: a review of toxicity. Arch Environ Occup Health. 2009;64(3):207-12. PubMed
- Brauner C, Joveleviths D, Álvares-da-Silva MR. Hepatotoxicity by methyl methacrylate. Clin Res Hepatol Gastroenterol. 2021;45(1):101513. PubMed
- Pemberton MA, Kimber I. Methyl methacrylate and respiratory sensitisation: a comprehensive review. Crit Rev Toxicol. 2022;52(2):139-166. PubMed
- Khanna G, Cernovsky J. Bone cement and the implications for anaesthesia. Vol 12. 4 ed. BJA Education: Oxford Academic; 2012:213-6. Link
- Donaldson AJ, Thomson HE, Harper NJ, Kenny NW. Bone cement implantation syndrome. Br J Anaesth. Jan 2009;102(1):12-22. PubMed
- Kinoshita H, Iranami H, Fujii K, et al. The use of bone cement induces an increase in serum astroglial S-100B protein in patients undergoing total knee arthroplasty. Anesth Analg. 2003;97(6):1657-60. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.