Copy link
Aortic Insufficiency: Etiology, Pathophysiology, and Clinical Presentation
Last updated: 01/04/2023
Key Points
- Aortic insufficiency (AI) is caused by the failure of the aortic valve leaflets to close.
- Bicuspid aortic valves and primary diseases of the ascending aorta and sinuses of Valsalva are the main causes of chronic and severe AI in high-income countries. Rheumatic heart disease is the main cause of AI in lower-income countries.1
- AI is typically a chronic disease in most patients, which results in left ventricle ventricular dilation and eccentric hypertrophy.1
Etiology
- AI occurs due to a failure of the aortic valve leaflets from fully closing (coapting) during diastole. The etiology of AI is broad and can be categorized in the following ways (Figure 1).
- Type 1: aortic root or aortic annular dilation
- Type 2: excessive aortic cusp motion
- Type 3: restricted leaflet motion or poor tissue quality leaflets
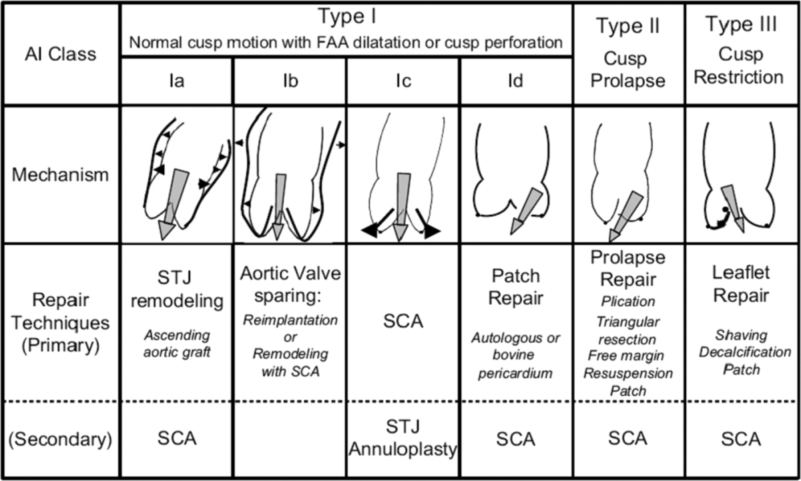
Figure 1. Functional classification of aortic insufficiency with a description of disease mechanisms and possible repair techniques. AI = aortic insufficiency; FAA = functional aortic annulus; STJ = sinotubular junction; SCA = subcommissural annuloplasty. Reproduced from Van Dyck MJ et al. Transesophageal echocardiographic evaluation during aortic valve repair surgery. Anesth Analg. 2010;111(1):59-70.
- The most common cause of chronic AI is congenital valvular disease (specifically bicuspid valve) and aortic dilation.1
- Causes of acute AI include endocarditis, aortic dissection, and prosthetic valve complications such as leaflet dysfunction, paravalvular leak, and valve dehiscence.2
- Other causes include age-related senile valvular calcification, age-related aortic dilation, systemic hypertension, myxomatous valve degeneration, aortitis, and aortic root dilation from syphilis or rheumatologic disease.2
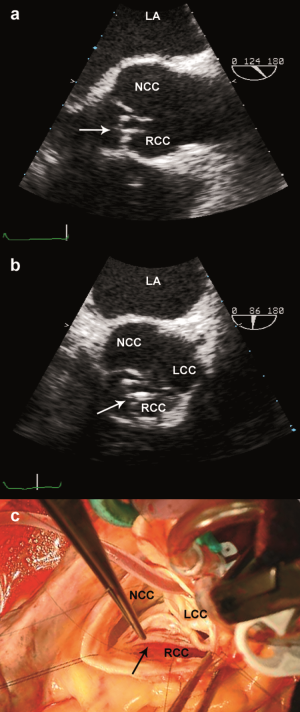
Figure 2. Thickened aortic valve leaflets. RCC = Right coronary cusp; NCC = Non-coronary cusp; LCC = Left coronary cusp. Reproduced from Van Dyck MJ et al. Transesophageal echocardiographic evaluation during aortic valve repair surgery. Anesth Analg. 2010;111(1):59-70.
Epidemiology
- Trace AI as seen on echocardiography with color Doppler is very common and is seen even in healthy patients.3
- AI prior to age 50 is unusual and when present, typically progresses over time.3 It is estimated to occur in approximately 2% of the population older than 70 years, with men affected more commonly than women.3
- Bicuspid aortic valves are estimated to affect 0.5-3% of the general population, with prevalence being three times higher in males than females.4 Of these patients, between 13-30% will develop moderate to severe AI in their lifetimes, and up to 40% of these patients will also develop an aortopathy with enlargement of the sinuses of Valsalva or ascending aorta.4
Pathophysiology
- AI produces a predictable hemodynamic response. When the aortic valve leaflets fail to coapt during diastole, a fraction of the left ventricle (LV) stroke volume leaks from the aorta back into the LV. This causes an increase in LV end-diastolic volume and LV wall stress. In response, the LV adapts to the increased volume through eccentric hypertrophy and dilation.
- Initially, the LV increases total stroke volume and maintains forward flow. Through eccentric hypertrophy and LV dilation, which maintains LV compliance, LV end-diastolic pressure initially remains normal despite the increase in LV volume. Low aortic diastolic blood pressures and a widened pulse pressure result from the increasing regurgitant volume back into the LV.
- As the disease progresses, there is an eventual increase in LV wall stress and afterload, which result in progressive LV systolic dysfunction in the setting of dilated cardiomyopathy.
- To compensate for a reduction in cardiac output, there is further peripheral vasoconstriction to maintain blood pressure. This adaptation eventually becomes pathologic, further exacerbating the severity of regurgitation in advanced stages of aortic insufficiency.5
Clinical Presentation
- Patients with chronic regurgitation may be asymptomatic for decades, even with evidence of ventricular remodeling.
- Those who develop severe disease have typical signs and symptoms of heart failure, including exertional dyspnea, angina, orthopnea, paroxysmal nocturnal dyspnea, and/or pulmonary edema. Of note, angina in these patients can be due to underlying atherosclerotic disease or poor coronary perfusion because of low diastolic aortic pressure.
- On auscultation, the classic decrescendo early diastolic murmur can be heard at the left upper sternal border.
- The pulse pressure is widened with a high systolic and low diastolic pressures.
- Bounding carotid pulses, head bobbing, retinal, and uvular pulsations are some of the peripheral signs of chronic AI.2
- Chest x-ray shows pulmonary congestion and cardiomegaly.
- Echocardiography is the gold standard in the evaluation of aortic regurgitation, providing valuable diagnostic information regarding severity, etiology, and pathologic maladaptations (Figure 3).
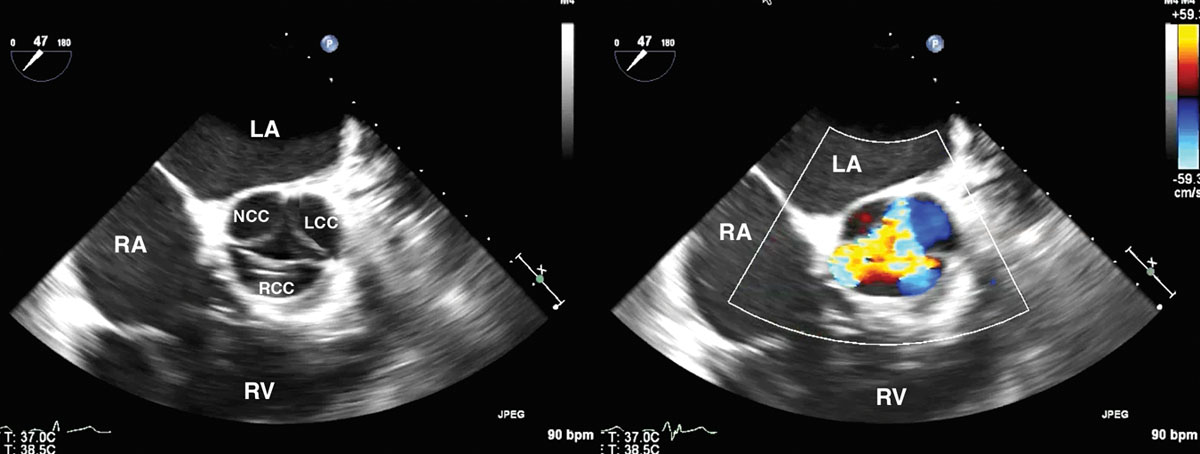
Figure 3. Aortic regurgitation in TEE midesophageal short-axis view with and without color flow Doppler. LCC = left coronary cusp; RCC = right coronary cusp; NCC = noncoronary cusp; LA = left atrium; RA = right atrium; RV = right ventricle. Reproduced from Von Homeyer P and Oxorn DC. Aortic Regurgitation: Echocardiographic Diagnosis. Anesth Analg. 2016;122(1):37-42.
- The American Heart Association (AHA) stages AI based on symptoms, valve anatomy, severity of valve dysfunction, and degree of cardiac maladaptation or hemodynamic effects.5
- Stage A: No or trace AI
- Stage B: Mild to moderate regurgitation with normal systolic function and only mildly dilated LV volume
- Stage C: Asymptomatic, severe regurgitation: Stage C1 is characterized by normal LV systolic function with mild to moderately increased LV dilation, while stage C2 is characterized by a decreased LV systolic function as well as markedly increased LV systolic volume.
- Stage D: Symptomatic, severe aortic regurgitation, along with decreased systolic function and severely increased LV volume
- Acute AI is less common than chronic regurgitation and is poorly tolerated because the LV has not had time to adapt to the increase in LV end-diastolic volume. In these settings, the LV typically fails quickly, rapidly resulting in pulmonary edema and hemodynamic instability unless the underlying cause of insufficiency is addressed.
See Also
Aortic Insufficiency: Hemodynamic Management, Medical and Surgical Management
References
- Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation. 2020; 143(5): e72-e227. PubMed
- Akinseye OA, Pathak A, Ibebuogu UN. Aortic valve regurgitation: A comprehensive review. Curr Probl Cardiol. 2018: 43(8): 315-34. PubMed
- Singh JP, Evans JC, Levy D, et al. Prevalence and clinical determinants of mitral, tricuspid, and aortic regurgitation (the Framingham Heart Study). Am J Cardiol. 1999: 83(6): 897-902. PubMed
- Masri A, Svensson LG, Griffin BP, et al. Contemporary natural history of bicuspid aortic valve disease: A systematic review. Heart. 2017; 103:1323-30. PubMed
Other References
- Zoghbi WA. Clinical manifestations and diagnosis of chronic aortic regurgitation in adults. In Otto CM (Ed). UpToDate. 2022. Accessed October 17th, 2022. Link
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.