Copy link
Hyperbaric Oxygen Therapy
Last updated: 05/23/2023
Key Points
- Hyperbaric oxygen (HBO) therapy utilizes Henry’s Law (the amount of gas that dissolves in a liquid is directly proportional to its partial pressure) to increase the dissolved oxygen content (0.3 mL/dL at sea level [1 atm] vs. 6 mL/dL at 3 atm pressure) and oxygen delivery.
- Hyperbaric oxygen delivery results in a decrease in the minimum alveolar concentration (MAC) due to an increase in partial pressures of the volatile agent for a given concentration at higher barometric pressures.
- Hyperbaric oxygen-related seizures are more likely with long, sustained treatment sessions (90-120 minutes without short intervals of room air) and the use of near-maximal pressures (2.8-3 atm).
Overview & Indications
- HBO therapy is an adjunct therapy that can be used to treat a number of medical conditions (Table 1).1-3 HBO therapy utilizes Henry’s Law which states that at a constant temperature and at equilibrium, the amount of gas that dissolves in a liquid is directly proportional to its partial pressure. For instance, at sea level (1 atm), the dissolved plasma oxygen concentration is 0.3 mL/dL, whereas HBO delivered at 3 atm results in a dissolved plasma oxygen concentration of 6 mL/dL resulting in increased oxygen delivery to end organs and tissues.2-3
- The indications for HBO therapy can be grouped into three main categories (Table 1).2,3
1. Wound healing acceleration and angiogenesis enhancement
2. Antimicrobial effects
3. Medical emergencies.
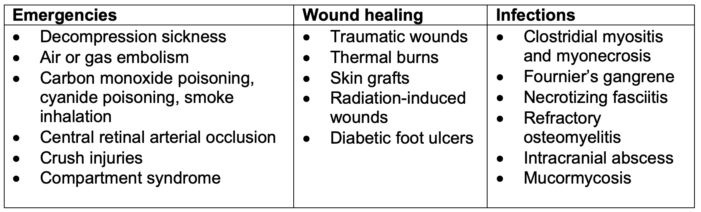
Table 1. Indications for HBO therapy
-
- Newer indications include:
- COVID-19 pneumonia and other hypoxic respiratory failures4
- An emerging anesthesia-specific indication is the treatment of perioperative nerve injuries5 as well as a potential use in the treatment and modulation of chronic pain.6
- Newer indications include:

Figure 2. Mobile pressure box for hyperbaric oxygen therapy (Public Domain, via Wikimedia Commons) Link.
Mechanism of Action
- As mentioned earlier, based on Henry’s law, HBO therapy increases oxygen content and delivery in conditions such as profound anemia, carbon monoxide poisoning, acute and chronic ischemia.
- In addition to Henry’s Law, HBO therapy involves the discussion of several other gas laws, particularly when discussing potential anesthetic effects. These include:
- Boyle’s Law: at a constant temperature, the volume of a gas is inversely proportional to the pressure exerted on it. This explains the role of HBO therapy in reducing the size of nitrogen bubbles in decompression sickness and gas embolism. At 3.0 atm, the size of nitrogen bubbles decreases by about two-thirds.
- Gay-Lussac’s Law: at constant volume, the pressure of a gas is proportional to its temperature.
- Charles’ Law: at constant pressure, the volume of a gas will vary directly with its temperature.
- Dalton’s Law: the total pressure of a mixture of gases is equal to the sum of their partial pressures.
- The effects of HBO therapy on improved wound healing are mediated by the generation of reactive oxygen species (e.g., oxygen-derived free radicals, hydrogen peroxide) and reactive nitrogen species (nitric oxide) in the tissues.2,3 HBO-mediated hyperoxia causes vasoconstriction which reduces vasogenic edema. HBO also reduces ischemia-reperfusion-induced leukocyte influx.
- Volatile agents produce anesthesia based on the partial pressure of the anesthetic in the body, not on the volume of the agent in the lungs. When volatile agents are administered to patients undergoing HBO therapy, this causes a decrease in the MAC because there is an increase in partial pressure of the volatile agent for a given concentration at higher barometric pressures. There is also an increase in gas density in HBO therapy, which also causes the rotameter flowmeters to read falsely high. For example, 2.0% sevoflurane at 1 atm would produce the same amount of anesthesia as 0.66% sevoflurane at 3 atm.
Complications
- Common side effects of HBO include claustrophobia, fatigue, headache, and vomiting. More serious complications include barotrauma (middle ear, sinus, pulmonary), pulmonary oxygen toxicity, reversible myopia, and central nervous system (CNS) oxygen toxicity (i.e., seizures).
- Seizures during HBO therapy are quite rare (~1 in 50,000 patients).7
- Risk factors include prior history of seizures or head trauma as well as those receiving glucocorticoids, insulin, thyroid replacement, and sympathomimetic medications.
- Seizure risk is increased when patients are exposed to HBO for greater than 90-120 minutes and when using pressures of 2.8-3 atm.
- Management of CNS oxygen toxicity seizures includes7
- terminating the HBO therapy session;
- decreasing the FiO2 to 0.21;
- anticonvulsant therapy; and
- supportive care.
Contraindications
- The only absolute contraindication to HBO therapy is untreated pneumothorax. Relative contraindications for HBO therapy include2,3
- pacemaker and other implanted devices;
- pregnancy;
- severe chronic obstructive pulmonary disease, asthma;
- epilepsy;
- hypoglycemia;
- current fever; and
- concurrent treatment with bleomycin (oxygen toxicity) or doxorubicin (synergistic cardiotoxicity).
References
- Leach RM, Rees PJ, Wilmshurst P. Hyperbaric oxygen therapy. BMJ. 1998; 317:1140-3. PubMed
- Ortega MA, Fraile-Martinez O, García-Montero C, et al. A General overview on the hyperbaric oxygen therapy: Applications, mechanisms, and translational opportunities. Medicina. 2021; 57(9):864. PubMed
- Mechem CC, Manaker S. Hyperbaric oxygen therapy. In: Post T (ed). UpToDate; 2023. Accessed March 7th, 2023.
- Boet S, Etherington C, Ghanmi N, et al. Efficacy and safety of hyperbaric oxygen treatment to treat COVID-19 pneumonia: a living systematic review update. Diving Hyperb Med. 2022;52(2):126-35. PubMed
- Brenna CT, Khan S, Katznelson R, et al. The role of hyperbaric oxygen therapy in the management of perioperative peripheral nerve injury: a scoping review of the literature. Reg Anesth Pain Med. 2022; rapm-2022-104113. PubMed
- Sutherland AM, Clarke HA, Katz J, et al. Hyperbaric oxygen therapy: A new treatment for chronic pain? Pain Pract. 2016;16(5):620-8. PubMed
- Hadanny A, Meir O, Bechor Y, et al. Seizures during hyperbaric oxygen therapy: retrospective analysis of 62,614 treatment sessions. Undersea Hyperb Med. 2016;43(1):21-8. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
