Copy link
Evoked Potentials: Modalities and Anesthetic Effects
Last updated: 01/05/2023
Key Points
- Intraoperative neurophysiologic monitoring (IONM) aims to protect the integrity of the peripheral and central nervous systems during surgical manipulation.1-3
- Anesthetic agents affect neuronal excitability by altering the synaptic function and thereby affecting monitored signals to variable degrees.2,3
- In general, monitored evoked responses are more sensitive to inhalational agents and nitrous oxide than intravenous agents, and cortical waveforms are more affected than subcortical or peripheral waveforms.2
- IONM modalities affected by anesthetics from most sensitive to least sensitive are motor evoked potentials (MEP) > electroencephalography (EEG) / somatosensory evoked potentials (SSEP) > brainstem auditory evoked potentials (BAEP).2,3
Introduction
- IONM is commonly used during surgery to protect the integrity of the peripheral and central nervous systems and reduce neurological complications.1,3
- The surgeon and monitoring team typically choose the most relevant modalities, and the monitoring team alerts the surgeon and anesthesiologist of signal change and impending injury. This allows real-time modification in the management and prevention of permanent injury.2,4
- The most used modalities include SSEPs, MEPs, EEG, BAEPs, and electromyography (EMG)/nerve conduction studies.1-3
Somatosensory Evoked Potentials
- SSEPs monitor the sensory pathway (through the dorsal root ganglia and posterior column) by electrical stimulation of a peripheral nerve distal to surgery and recording the potential at different landmarks as it propagates along the pathway from the peripheral nerves, through the spinal cord, subcortical and cortical regions (Figures 1 and 2).1-5
- Typically, the ulnar or median nerve is stimulated for the upper extremity and the posterior tibial nerve for the lower extremity.2
- SSEPs are sensitive to all inhalational agents and nitrous oxide.
- SSEPs are less affected by intravenous agents. Ketamine and etomidate increase SSEP amplitude.2
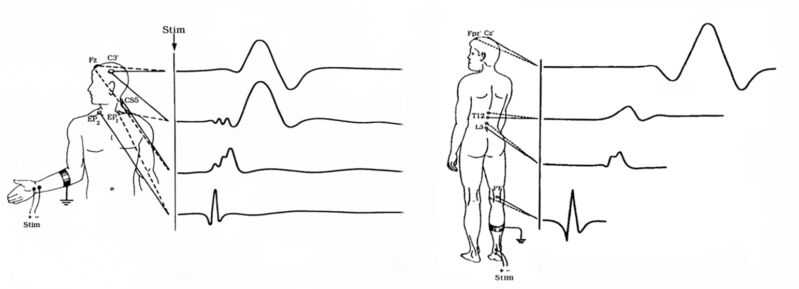
Figure 1. Upper and lower extremity SSEPs. Source: Neurophyspedia, the Wikipedia of Intraoperative Neurophysiology. (CC BY-SA 3.0)5
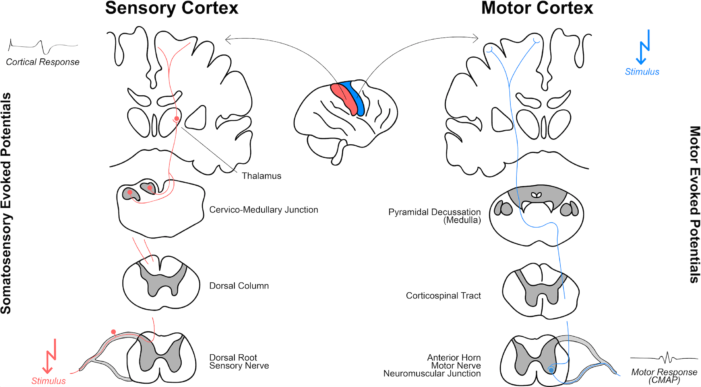
Figure 2. Principles of SSEPs and MEPs. Reproduced from Maier S, Goebel U, Krause S, et al. Somatosensory and transcranial motor evoked potential monitoring. PLoS ONE. 2018. 13(10): e0205410. CC-BY 4.0.6
- The more synapses that participate in generating the signal, the bigger the effect of anesthetic agents. For example, in the cortex, many more synapses are responsible for the cortical waveforms, which is why cortical waveforms are more affected by anesthetics.
- Affected waveforms: Cortical > subcortical > peripheral response.2
Motor Evoked Potentials
- MEPs monitor the motor pathway (including the motor cortex, corticospinal tract, nerve root, and peripheral nerve) by transcranial electric, and less commonly magnetic stimulation of the motor cortex, or direct stimulation of the brain surface, and recording the elicited muscle contractions from corresponding muscles by recording electrodes (Figures 2 and 3).2,3
- MEP compliments but does not replace SSEP.
- MEPs are more sensitive to the effects of anesthetic agents than SSEPs, and MEP changes precede changes in SSEP, allowing time to react.2
- MEPs are affected by inhalational more than intravenous agents. MEPs are most susceptible to anesthetics agents at the motor cortex and anterior horn cells (alpha motor neurons and interneurons).1-2
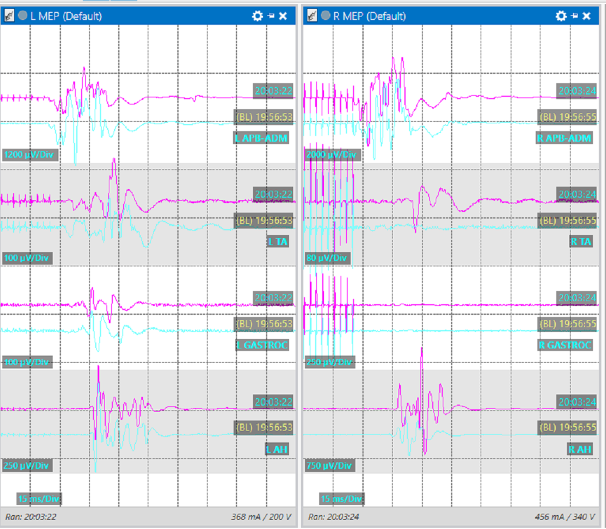
Figure 3. MEP response from various muscle groups.
Brainstem Auditory Evoked Potentials
- BAEPs monitor the auditory nerve and auditory pathway through the brainstem by acoustic stimulation of the auditory nerve and scalp surface electrode recording (Figure 4). Waves I-V are typically monitored. Characteristic changes are prolonged interpeak latencies of waves I-III and waves I-V.4
- The brainstem portion of the auditory pathway is made up of a relatively small number of neurons with very little contribution from interneurons, which makes this pathway robust and highly resistant to anesthetic effects.4
- BAEPs are used mostly during base of skull surgery.
- Anesthetic agents, including inhalational agents and neuromuscular blockers (NMBA), have little effect on BAEPs.
- BAEPs may be affected by hypothermia.
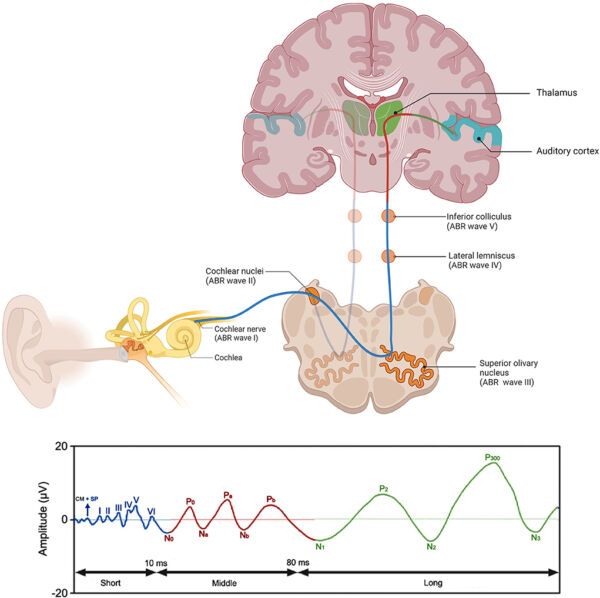
Figure 4. Pathway for BAEP. From Jacxsens L, et al. Brainstem evoked auditory potentials in tinnitus: A best evidence synthesis and meta-analysis. Front. Neuro. 2022; 13:941876. CC BY 3.07
Electroencephalography
- EEG monitors cortical electrical activity with surface scalp electrodes or brain surface electrodes to detect cortical ischemia or seizure activity.2
- EEG can also be used to monitor the depth of anesthesia.
- Surface scalp electrodes are used to evaluate cerebral perfusion during carotid endarterectomy and to detect seizure activity during intracranial surgeries or during MEP monitoring.
- Brain surface electrodes are used during seizure surgery (electrocorticography or ECoG), brain mapping, and select tumor resections.
- The effect of anesthetics on EEG is dose-dependent and similar to their effect on cortical evoked potentials. EEGs are moderately sensitive to inhalational agents and insensitive to NMBA.2
- EEG changes often include an initial increase in frequency, followed by slowing, asymmetry, and ultimately electrical silence (isoelectricity).2
Electromyography/Nerve Conduction Studies
- These modalities monitor nerve roots and peripheral nerves by detecting muscle activity (EMG) or nerve action potentials (NCS).
- Spontaneous or free-run EMG provides continuous monitoring and detection of neuronal structures during surgical dissection around nerve roots, cranial, and peripheral nerves.
- Triggered EMG is used to identify and map nerve roots and nerves by delivering stimulation intermittently. It can be used for assessment of pedicle breach during pedicle screw placement.
- During EMG and NCS, there are no synapses between the stimulating and recording site, making it less sensitive to the effects of anesthetic agents.2
- Except for NMBAs, anesthetic agents have no significant effects on EMG.
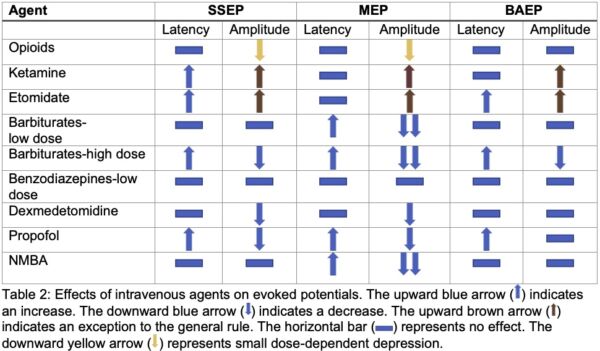
Effects of Anesthetics Agents on IONM1
Inhalational agents
- Halogenated agents (e.g., isoflurane, sevoflurane, desflurane):
- Increased latency: MEP > SSEP; minimally for BAEP
- Decrease amplitude: MEP > SSEP; minimally for BAEP
- Nitrous oxide (N2O):
- Increase latency: MEP > SSEP; minimally for BAEP
- Decrease amplitude: MEP > SSEP > BAEP

Intravenous Agents
- Lesser effect on monitoring than inhalational agents
- SSEP
- Increased latency and decreased amplitude: propofol and high dose barbiturates
- Increased amplitude: ketamine and etomidate
- Little effect: opioids and benzodiazepines
- MEP
- Increased latency and decreased amplitude: propofol, barbiturates, dexmedetomidine in higher doses, and lidocaine
- Little to no effect: opioids, ketamine, and etomidate
- BAEP: may increase latency (high dose barbiturates, otherwise little to no change)
- SSEP
Neuromuscular Blocking Agents
• Affects EMG and MEP; no effect on SSEP, BAEP, EEG
• Low-dose continuous infusion of NMBAs controlled with train-of-four monitoring may be used when muscle relaxation is desired (e.g., during surgical exposure). However, this reduces the accuracy of motor monitoring and is not used routinely.
References
- Husain, A. Practical Approach to Neurophysiologic Intraoperative Monitoring. Demos Medical Publishing, 2008. ProQuest Ebook Central.
- Koht A, Sloan TB, Toleikis JR (Editors) Monitoring the Nervous System for Anesthesiologists and Other Health Care Professionals, Second Edition. Springer, 2017.
- Shils JL, Sloan TB. Intraoperative neuromonitoring. Int Anesthesiol Clin. 2015 Winter;53(1):53-73. PubMed
- Legatt AD, Arezzo JC, Vaughan HG Jr. The anatomic and physiologic bases of brain stem auditory evoked potentials. Neurol Clin. 1988;6(4):681-704. PubMed
- Maier S, Goebel U, Krause S, et al. Somatosensory and transcranial motor evoked potential monitoring. PLoS One. 201813(10): e0205410. PubMed
- Jacxsens L, De Pauw J, Cardon E, et al. Brainstem evoked auditory potentials in tinnitus: A best evidence synthesis and meta-analysis. Front Neuro. 2022; 13:941876. PubMed
Other References
- Neurophyspedia, the Wikipedia of Intraoperative Neurophysiology. Somatosensory Evoked Potentials. Published October 25, 2013. Updated January 18, 2022. Accessed November 10, 2022. Link
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.