Copy link
Blood-Brain Barrier
Last updated: 04/03/2023
Key Points
- The blood-brain barrier (BBB) is a barrier that prevents most compounds from traversing from the blood to the brain to create a separate, protected environment in the central nervous system (CNS) for optimal neuronal function.
- BBB dysfunction contributes to pathology in several neurological diseases, including tumors, traumatic brain injury, degenerative diseases, epilepsy, and intracranial hypertension.
- The effectiveness of hyperosmolar therapy to facilitate brain relaxation or decrease intracranial pressure depends on the integrity of the BBB.
- Solutes administered in high osmolarity fluids can’t cross the BBB and therefore can draw water out of the brain parenchyma. High osmolarity solutes don’t have this effect on most other organs because they can freely cross the vessel wall.
Anatomy
- The BBB is a cellular barrier that tightly controls the microenvironment of the CNS to allow for proper neuronal function.1
- Physical components of the BBB include (Figure 1):1
- Endothelial cells
- Basement membrane
- Pericytes
- Perivascular fluid space (Virchow-Robin space)
- Astrocyte end-feet
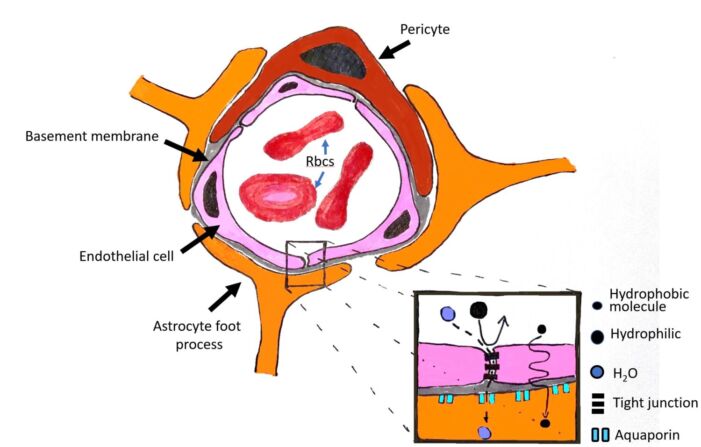
Figure 1. Components of the BBB. The BBB is composed of cerebral capillary wall endothelial cells that are joined by tight junctions. Pericytes and astrocyte foot processes reinforce the endothelial cell barrier and collectively form the BBB. Insert shows that the tight junctions form a barrier to larger molecules (large black circles) but not to water (blue circles). Small lipophilic molecules (small black circles) can also passively diffuse.
- The absence of fenestrations is another characteristic feature of the BBB.
- Tight junctions (TJs) are present between endothelial cells to prevent blood-borne substances from entering the CNS through passive paracellular diffusion. They consist of 3 integral membrane proteins – occludin, claudin, and junction adhesion molecules.1
- Astrocytes protect the BBB against hypoxia-induced disruption of TJs and thus, the permeability of the BBB.
- Areas of the brain not protected by the BBB include1
- Area postrema
- Median eminence of the hypothalamus
- Pineal gland
- Posterior pituitary
- Preoptic pecess
- Choroid plexus
Transport Function of the BBB
- Several mechanisms for transport across the BBB have been proposed.2

- Solutes that easily traverse the BBB have the following properties:
- Small molecular weight
- Uncharged
- Lipophilic
- Substrate for active transport
- High concentration in the bloodstream
- Low protein binding
- Molecular similarity with another actively transported substrate
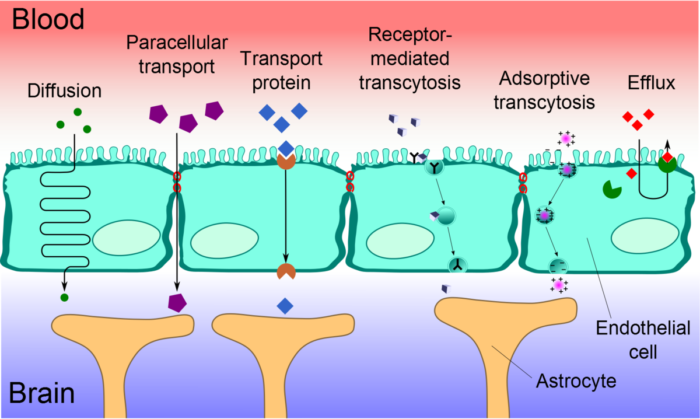
Figure 2. Transport mechanisms across the BBB. Source: Wikimedia Commons. User:Kuebi= Armin Kübelbeck, CC BY 3.0 Link.
Hyperosmolar Therapy
- Hyperosmolar therapy may be used to decrease brain volume electively (e.g., to facilitate surgical exposure) or emergently (e.g., to prevent impending herniation).
- Mechanism of action of hypertonic saline and mannitol.3

- Caution is advised for administering hyperosmolar therapy when there is a suspicion of BBB breakdown, as hyperosmolar therapy may worsen edema. Many conditions that cause elevated ICP lead to dangerous feedback loops that causes even more edema: breakdown of BBB → edema → elevated intracranial pressure (ICP) → ischemia → further breakdown of the BBB.
Pathophysiology
- The integrity of tight junctions can be compromised in a variety of pathological conditions through different pathways (Figure 3).3
- Pathological processes commonly involve the production and release of inflammatory mediators such as bradykinin, histamine, substance P, free radicals, glutamate, endothelin-1, etc.
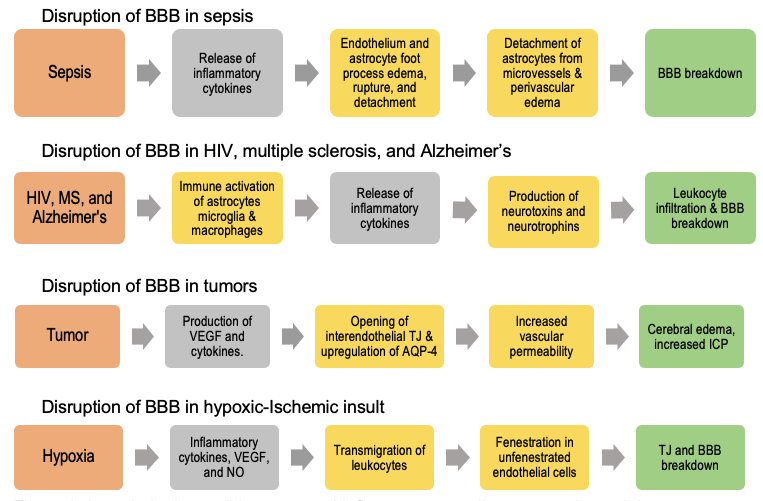
Figure 3. In pathologic conditions, several inflammatory mediators are released that compromise BBB integrity.3 Orange boxes indicate diseases, grey boxes indicate mediators of pathological processes, yellow boxes indicate molecular and cellular pathological mechanisms, and green boxes represent outcomes. (HIV: human immunodeficiency virus; MS: multiple sclerosis; BBB: blood-brain barrier; VEGF: vascular endothelial growth factor; TJ: tight junction; AQP-4: aquaporin-4; ICP: intracranial pressure; NO: nitric oxide).
Clinical Implications
- Increased risk for elevated ICP
- Drugs that cross the BBB via diffusion are small, lipophilic, noncharged molecules (e.g., atropine, scopolamine) and will thus cause CNS effects.
- Compromise of the BBB integrity contributes to the pathological process of several neurological problems.
References
- Daneman R, Prat A. The blood-brain barrier. Cold Spring Harb Perspect Biol. 2015;7(1): a020412. PubMed
- Abbott NJ, Patabendige AA, Dolman DE, et al. Structure and function of the blood-brain barrier. Neurobiol Dis. 2010;37(1):13-25. PubMed
- Ballabh P, Braun A, Nedergaard M. The blood-brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol Dis. 2004;16(1):1-13. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.