Copy link
Difficult Airway Management in Trauma Patients
Last updated: 01/12/2023
Key Points
- Difficult airways in trauma patients do not follow the standard ASA difficult airway algorithm.
- Surgical airway may be the best initial choice in airway management.
- Awake fiberoptic intubation (AFOI) should be considered earlier but is highly dependent on patient factors (cooperativeness, hemodynamics) in an emergency situation.
Introduction
Trauma airway management poses unique challenges to even the most skilled physician, which is why trauma anesthesiologists are invested in expanding research, education and developing trauma airway guidelines. In 2005, William Wilson outlined modifications to the American Society of Anesthesiologists (ASA) Difficult Airway (DA) algorithm for trauma patients and proposed specific algorithms for several trauma scenarios.1 More recently, the ASA Committee on Trauma and Emergency Preparedness (COTEP) updated their recommendations for difficult airway management in trauma patients.
Over the last decade, trauma airway management techniques have expanded to include video assisted laryngoscopy (VAL), supraglottic airway (SGA) devices, cricothyrotomy, and others. However, there remains insufficient data to demonstrate an advantage of any modality over another. Despite this, useful recommendations and guidance were made based on current scientific literature and expert opinion.2
Trauma Airway Challenges
Advanced Trauma Life Support (ATLS) defines a definitive airway as a cuffed endotracheal tube, achieved either by orotracheal intubation, nasotracheal intubation, cricothyrotomy, or tracheostomy.
Trauma patients pose several unique challenges for airway management.2
- Limited time to secure a definitive airway
- Difficulty recognizing a difficult airway
- Inability to wake the patient up and cancel the case if airway issues are encountered
- Hemodynamic instability in concert with single or mixed shock states
- Altered head, neck, and airway anatomy
- Risk of aspiration
- Cervical spine immobilization and need for cervical spine protection
- Uncooperative patient
- Rapidly evolving and possibly competing clinical priorities
- Problems with positioning
- Trauma team dynamics and experience
- Prehospital scene safety
- Triage, equipment, and mass casualty possibilities
- Chemical, biological, radiological, and nuclear exposures.
Induction Agents and Muscle Relaxants
- Selecting an induction agent poses additional challenges in the trauma patient.3 These drugs may cause hypotension through peripheral vasodilation and myocardial depression. Thus, lower doses and careful titration of agents is often necessary while aiming to provide adequate hypnosis, amnesia, and muscle relaxation. Furthermore, given the insufficient nil per os (NPO) status and delayed gastric emptying in trauma patients, rapid sequence intubation (RSI) is commonly used. Interestingly, ATLS-10 has replaced the term RSI with “drug-assisted” intubation.
- Slower-sequence intubation, which involves the slower titration of induction agents may be considered in trauma patients with difficult airways, patients not tolerating preoxygenation, hemodynamic instability, etc.3
- An ideal induction agent should have the following properties:
- rapid onset;
- minimal adverse effects (hemodynamic instability);
- improves intubating conditions; and
- provides sedation and decrease awareness.
Commonly used induction agents in trauma patients are listed in Table 1.
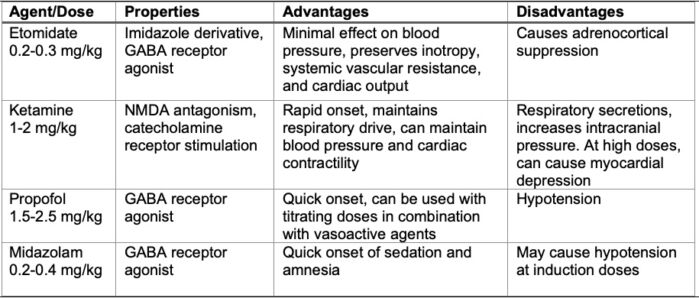
Table 1. Commonly used induction agents in trauma patients. (GABA = gamma aminobutyric acid, NMDA = N-methyl-D-aspartate)
- Neuromuscular blocking agents (NMBA) are typically administered at RSI doses. Succinylcholine (1.5-2 mg/kg) and rocuronium (1.2 mg/kg) are the most commonly used NMBAs in trauma patients.
Difficult Airway Management in Trauma
Several strategies differ from the ASA DA algorithm in trauma patients. The following circumstances should be considered when planning to manage a traumatic airway.2
- The patient cannot be awakened or the procedure cannot be cancelled as the need for an emergent airway will presumably remain.
- A surgical airway may be the best initial choice in certain conditions (e.g., extensive oromaxillofacial trauma or suspected tracheal disruption).
- The physician may fail to recognize a DA while evaluating the patient prior to induction of general anesthesia or the patient has an obvious DA but is too unstable or uncooperative for an awake fiberoptic intubation (e.g., children, developmental delay, intoxication, poisonings, or head injuries).
- If intubation attempts are unsuccessful, bag valve mask (BVM) ventilation with 100% oxygen should be initiated. If BVM is adequate, other modalities to secure the airway can be employed.
- If BVM ventilation is inadequate, SGA placement should be attempted with simultaneous preparation for a cricothyrotomy should SGA placement fail.
- Cricoid pressure (CP) should be applied until the airway is secured. However, CP should be modified or removed if it interferes with airway instrumentation since a secured airway takes precedence over the potential risk of aspiration.2
- Gentle BVM ventilation (at less than 20cm of H2O) prior to intubation may prove beneficial in patients who don’t tolerate preoxygenation or those with limited functional residual capacity (e.g., lung injury, obesity). After a failed intubation attempt, BVM ventilation must be established.2
- In patients with suspected cervical spine injury, manual in-line stabilization (MILS) of the cervical spine during intubation is recommended. However, MILS may worsen the laryngoscopic view leading to a delay or failure to achieve a secured airway. Like CP, MILS should be modified or removed if it interferes with airway instrumentation.
- VAL is routinely used in patients with suspected cervical spine injuries, resulting in reduced cervical motion with laryngoscopy. Limitations for VAL include facial trauma causing a jaw-lock, limited mouth opening, or blood and secretions in the oropharynx impairing the laryngoscopic view. Of note, the use of VAL prolongs intubation times but has not impacted first-pass success rate nor survival to hospital discharge. Thus, VAL has not replaced DL in the trauma setting.
DA Algorithm in Trauma Patients
- The likelihood of problems with consent/cooperation, BVM ventilation, SGA placement, DL, or surgical airway access should be assessed.
- The basic ASA standards of monitoring should be adhered to.
- Supplemental oxygen should be delivered throughout the intubation attempt, if possible.
- The best airway management approach should be considered (awake fiberoptic intubation (AFOI), DL, VAL, invasive techniques, preserving or ablation of spontaneous respirations, etc.).
- Primary and alternative airway management plans should be developed (Figure 1).2
- The latest 2022 ASA practice guidelines for management of the difficult airway specifically emphasizes limiting the number of intubation attempts, calling for help early, and being aware of the passage of time during difficult airway management.4
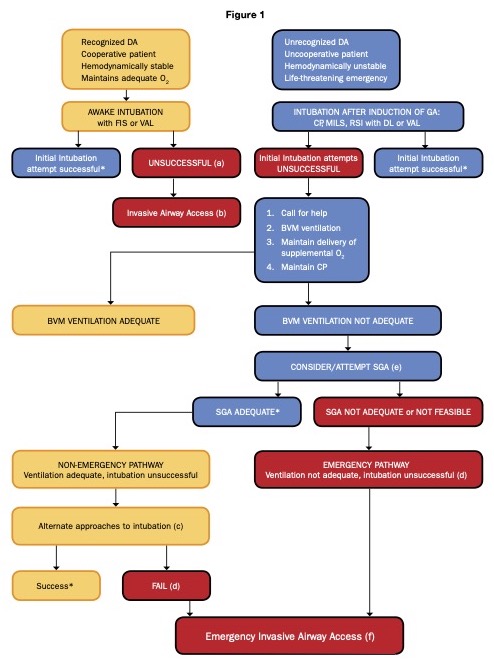
Figure 1. Difficult airway algorithm in trauma patients. Reproduced with permission from Hagberg CA, Kaslow O. Difficult airway management algorithm in trauma: Updated by COTEP. ASA Newsletter. 2014; 78:56–60.2
Footnotes.2
- Attempt BVM ventilation or SGA placement
- Options for invasive airway include surgical or percutaneous cricothyrotomy or tracheostomy, transtracheal jet ventilation, retrograde intubation, etc.
- Alternative approaches include VAL, SGA as a conduit for fiberoptic intubation, intubating stylet, light wand, etc.
- Awakening patient is often impractical in the trauma setting.
- Emergency non-invasive airway access includes SGA placement.
- A surgical airway kit should be immediately available.
References
- Wilson, WC. Trauma: airway management. ASA Monitor. 2005;69 (11): 9-16. Link
- Hagberg CA, Kaslow O. Difficult airway management algorithm in trauma: Updated by COTEP. ASA Newsletter. 2014; 78:56–60. Link
- Estime S, Kuza C. Trauma airway management: Induction agents, rapid versus slow sequence intubation, and special considerations. Anesthesiology Clin. 2019; 37:33-50. PubMed
- Apfelbaum JL, Hagberg CA, Connis RT, et al. 2022 American Society of Anesthesiologists practice guidelines for management of the difficult airway. Anesthesiology. 2022; 136(1):31-81. PubMed
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.