Copy link
Opioid Rotation and Equianalgesic Dosing
Last updated: 05/24/2023
Key Points
- Conversion of opioids from intravenous to oral formulation can be easily calculated utilizing equianalgesic dosing charts.
- When converting from one oral opioid formulation to another, incomplete cross-tolerance between the opioids must be accounted for and the dose reduced accordingly.
- Equianalgesic conversion ratio for intravenous to epidural/intrathecal varies, depending on the meningeal permeability of the opioid.
Opioid Rotation
- Opioid rotation is a strategy that involves the switch from one opioid to another in an effort to improve analgesia or to decrease opioid-related side effects.1,2
- When a patient demonstrates suboptimal analgesia despite appropriate opioid dosing, this strategy is commonly utilized.
- Switching opioids must be done carefully with appropriate calculation of drug doses to avoid withdrawal or toxicity, utilizing an equianalgesic dosing table (Table 1).
- Close monitoring is essential to ensure that the rotation is effective and pain has decreased with the medication changes.
Equianalgesic Dosing of Opioids: Intravenous to Oral
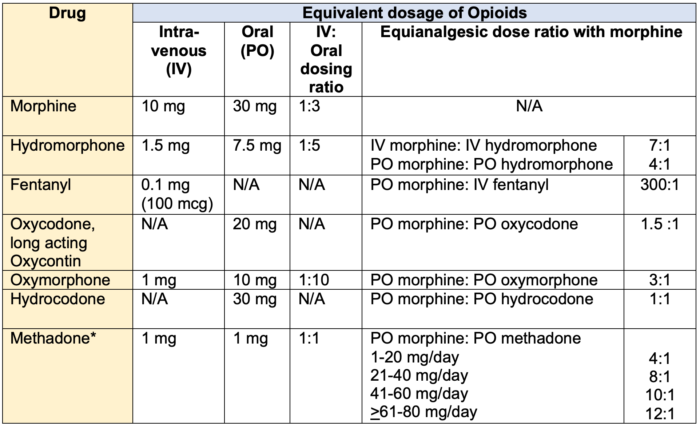
Table 1. Equianalgesic Dosing of Opioids: Intravenous to Oral.3 *Methadone: conversion factor increases at higher doses
Steps For Converting Opioids3-5
- The total 24-hour dose of the current opioid should be calculated.
- The opioid equivalence chart should be used to estimate the equivalent 24-hour dose of the new opioid in morphine milligram equivalents (MME).
- The dose of the new opioid should be reduced to help account for possible incomplete cross‐tolerance between opioids.
- If the previous opioid dose was 75 mg oral morphine equivalents or lower per 24 hours, reduce the dose of the new opioid to 60% to 75% of the previous opioid.
- If previous opioid dose was more than 75 mg oral morphine equivalents per 24 hours, reduce the dose of the new opioid to 50% or less of the previous opioid.
- The 24-hour dose of the new opioid should be divided based on the specific drug and dosage form selected.
Important Notes Regarding Opioid Conversion
- The doses included here provide a starting point for the converting different opioids during maintenance therapy; these are not the recommended doses for the initiation of opioid therapy.
- Equianalgesic conversions serve only as a general guide to estimate opioid dose equivalents.
- When switching to a new opioid, the initial daily dose for the new opioid determined by using the conversions in the first part of this table should be further reduced by 25 to 50% to adjust for lack of complete mu receptor cross-tolerance (except when switching to methadone, which requires a 75 to >90% reduction).
- In contrast, when switching between intravenous and oral administration of the same opioid, an empiric reduction of the equianalgesic dose estimate shown above is generally not necessary.
Equianalgesic Dosing of Opioids in Neuraxial Routes
- Equianalgesic conversion ratio for intravenous to epidural/intrathecal varies, depending on the meningeal permeability of the opioid (Table 2).
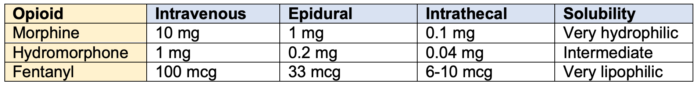
Table 2. Equianalgesic Dosing of Opioids in Neuraxial Routes
References
- Fine PG, Portenoy RK. Establishing “best practices” for opioid rotation: Conclusions of an expert panel. J Pain Symptom Manage. 2009;38(3):418-25. PubMed
- Knotkova H, Fine PG, Portenoy RK. Opioid rotation: the science and the limitations of the equianalgesic dose table. J Pain Symptom Manage. 2009;38(3): 426. PubMed
- Rosenquist R. Use of opioids in the management of chronic non-cancer pain. Post TW, ed. UpToDate. Waltham, MA: UpToDate Inc. Accessed on 3/21/2023. Link
- Centers for Disease Control and Prevention. Calculating total daily dose of opioids for safer dosage. Accessed 12/01/2022. Link
- British Columbia Provincial Academic Detailing Service. Opioids in chronic non-cancer pain: The basics. Updated 07/01/2013. Accessed on 12/05/2022. Link
Copyright Information

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.